1
A 2-substituted 8-hydroxyquinoline stimulates neural stem cell proliferation by
modulating ROS signalling
Cathryn L. Haigh,
1,
* Carolin Tumpach,
2
Steven J. Collins,
1
and Simon C. Drew
2,
*
1
Department of Medicine, Royal Melbourne Hospital, The University of Melbourne, Victoria
3010, Australia,
2
The Florey Department of Neuroscience and Mental Health, The University
of Melbourne, Victoria 3010, Australia.
* To whom correspondence should be addressed: sdrew@unimelb.edu.au (+61 3 9035 8684),
chaigh@unimelb.edu.au (+61 3 8344 1952)
Running Title:
8HQ stimulation of NSC proliferation
Keywords:
8-hydroxyquinoline, neural stem cells, neurosphere, proliferation, NADPH oxidase, reactive
oxygen species, 2-[(dimethylamino)methyl]-8-hydroxyquinoline, DMAMQ, PBT2

2
Abstract
Eight-hydroxyquinolines (8HQs) are a class of compounds that have been identified as
potential therapeutics for a number of neurodegenerative diseases. Understanding the
influence of structural modifications to the 8HQ scaffold on cellular behaviour will aid the
identification of compounds that might be effective in treating dementias. In this study, we
describe the action of 2-[(dimethylamino)methyl]-8-hydroxyquinoline (DMAMQ) on adult
murine neural stem cells (NSCs) cultured in vitro. Treatment of NSCs with DMAMQ
resulted in enhanced self-renewal and increased neurite outgrowth. Concurrent with the
positive growth effects was an increase in intra-cellular reactive oxygen species, with the
growth being inhibited by inactivation of the NADPH oxidase (Nox) enzyme family. Our
results indicate that DMAMQ can stimulate neurogenesis via the Nox signalling pathway,
which may provide therapeutic benefit in treating dementias of various types by replenishing
The narrow concentration range over which these
effects were observed, however, suggests there may exist only a small therapeutic window for
neuro-regenerative applications.

3
Introduction
Endogenous neural stem cells (NSCs) are an as-yet untapped resource in combatting
dementia. Low levels of NSCs persist in the brain throughout adult life, maintaining the
ability to self-replicate and to differentiate into mature brain cells. During dementia,
neurogenesis (the ability to form new neurones) is seen to become dysregulated [
1
,
2
]. In
rodent models of Alzheimer's disease (AD), production of beta-amyloid, one hallmark of AD,
changes the growth and differentiation of these cells [
3
,
4
,
5
,
6
,
7
]. Neurogenesis has been
extensively linked with both learning and forgetting [
8
,
9
,
10
]; therefore, changes in these
processes might contribute significantly to the cognitive changes that occur during
neurodegenerative diseases, including AD. Compounds that modulate or normalise the
functions of NSCs represent a prime target for alleviating the symptoms associated with, or
delaying the course of, neurodegenerative diseases.
Unbiased chemical screening of substituted 8-hydroxyquinolines (8HQs) and a range of
investigations in yeast, nematodes, mice and humans have highlighted the variable
mechanisms by they may act. Side-chain modifications to the 8HQ backbone may lead to
functional differences in vivo, with applications of substituted 8HQs as anti-microbial agents
[
11
,
12
], anti-cancer agents [
13
,
14
], epigenetic modulators [
15
,
16
], dementia treatments
[
17
,
18
,
19
,
20
], artificial nucleobases [
21
] and medical imaging agents [
22
,
23
]. Moreover,
biosynthesis of substituted 8HQs has been identified in mammals (eg. xanthurenic acid [
24
]),
bacteria (eg. quinolobactin [
25
]) and insects (eg. 2-carboxy-8HQ [
26
]). In dementia
applications, studies utilising 5,7-dichloro-2-[(dimethylamino)methyl]-8-hydroxyquinoline
reported an increased neurite number following treatment of cultured PC12 cells and a
restoration of hippocampal dendritic spine density (but not number) in transgenic mouse
model of AD [18].

4
In the current study, we sought to further understand the action of this class of 8HQ by
treating adult murine NSC cultures with 2-[(dimethylamino)methyl]-8-hydroxyquinoline
(DMAMQ; Figure 1). We specifically assessed the ability of these cells to self-renew,
essential for maintaining their numbers in the brain throughout life, and to differentiate into
new CNS lineage cells. Our findings show that whilst neurogenesis is unchanged, NSCs
treated with DMAMQ demonstrate a dose-dependent increase in NSC proliferation and
increased neurite outgrowth during neurogenesis that was signalled by increased production
of reactive oxygen species (ROS) signalling intermediates by the NADPH oxidase (Nox)
enzyme family. These effects were only observed within a narrow concentration range,
suggesting a small therapeutic window for neuro-regenerative applications.

5
Materials and Methods:
Synthesis
2-[(dimethylamino)methyl]-8-hydroxyquinoline (DMAMQ) was synthesised as described
previously [
27
].
Cell culture
Murine NSCs were harvested from the brains of 6-8 week old Balb/c mice and grown without
modification as neurospheres in liquid culture as described previously [
28
].
DMAMQ treatments
DMAMQ was included in the culture medium at the concentrations indicated. For all assays,
the compound was added as a single treatment at the start of the assay and was not
replenished when the media was changed.
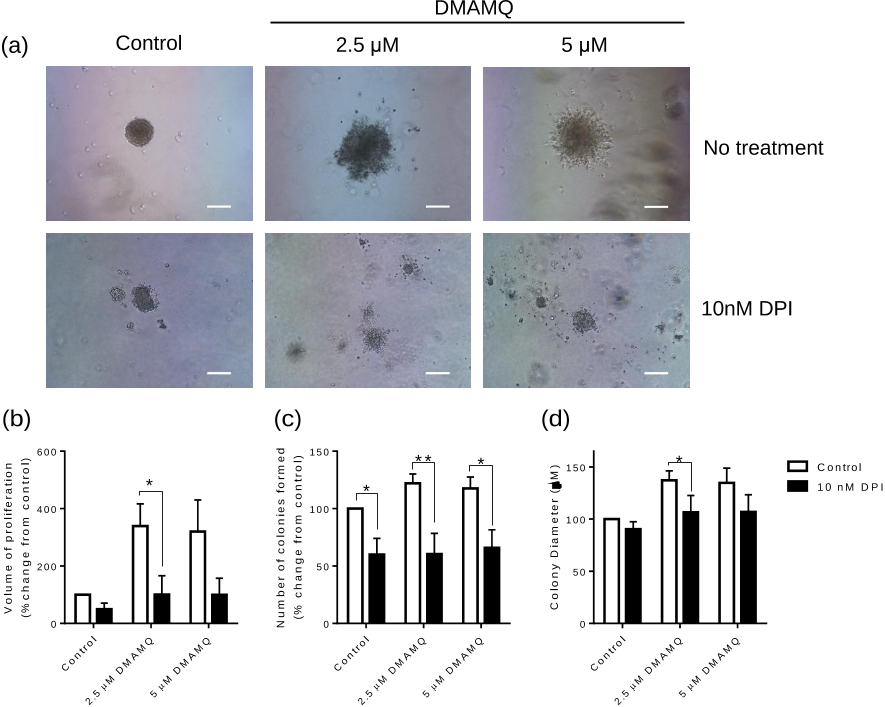
Neural Colony Forming Assay (NCFA)
The NCFA has been described previously [
29
]. For each independent experiment over 50
colonies were measured. As stated above, DMAMQ was included in the matrix once only at
the start of the incubation.
Neurite outgrowth assay
Neurite outgrowth was measured using a neurite outgrowth staining kit (Merck-Millipore) as
per the manufacturer's instructions with the following modifications. One hundred thousand
cells per condition were incubated in high FGF (20 ng/mL), no EGF, growth media before
![Figure 1. Structure of DMAMQ (2-[(dimethylamino)methyl]-8-hydroxyquinoline).](/figures/figure-1-structure-of-dmamq-2-dimethylamino-methyl-8-1jw2kicu.png)