Abstract: The effect of hydrogen and methane addition on the propagation and extinction of atmospheric CO/airflames was investigated experimentally and numerically. Experiments were conducted by using the counterflow, twin-flame technique and laser-Doppler velocimetry for the determination of laminar flame speeds and extinction strain rates. The simulation was conducted by using the one-dimensional flame code, and by solving the conservation equations of mass, momentum, energy, and species along the stagnation stream-line of the counterflow. In both cases, detailed description of the chemistry and transport was used. Results indicate that the addition of small amounts of hydrogen and methane to CO flames increases the laminar flame speeds and extinction strain rates by accelerating the main CO oxidation reaction. The sensitivity of the mass burning rate to this reaction is particularly high when trace amounts of hydrogen and methane are added. For large amounts of additives, the chemistry shifts toward that of the additive, and the advantages of the CO kinetic simplicity are lost. The experimental data were closely predicted by the numerical calculations for both propagation and extinction, indicating that existing CO, hydrogen, and methane kinetics can be used with confidence for similar studies. Detailed analysis of the flame structure revealed that, when CO and methane are both supplied as reactants, the CO oxidation follows that of methane, and that, for methane-rich mixtures, the supplied CO remains unreacted until the intermediate CO has been completely formed.
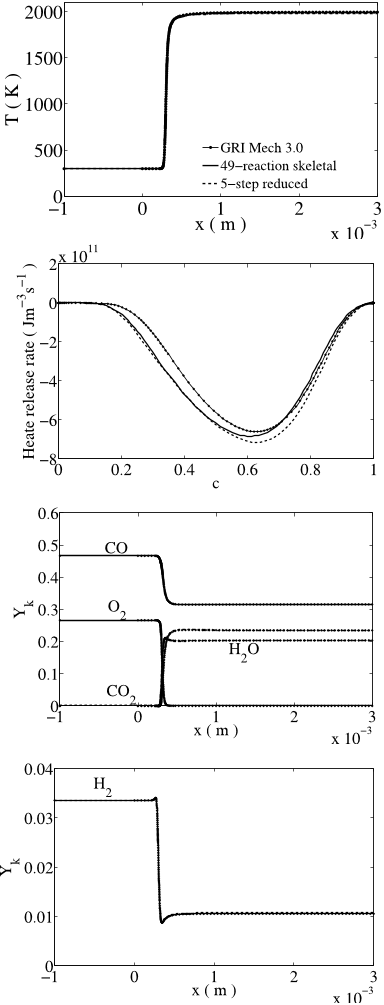
![Fig. 4: Laminar flame speeds of CO/H2-air mixtures using the reduced (dashed lines) and skeletal (full lines) mechanisms. Symbols: experimental results of Vagelopoulos and Egolfopoulos [48]. Tu = 298 K, p = 1 atm, XN2/XO2 = 3.76.](/figures/fig-4-laminar-flame-speeds-of-co-h2-air-mixtures-using-the-209eopff.png)
![Fig. 3: Laminar flame speeds of syngas mixtures ( CO/H2/CH4/CO2/N2-air ) using the reduced (dashed lines) and the skeletal (full lines) mechanisms. Symbols: experimental results of Yong et al. [27]. fCH4 = 0.24 with 11% CO2 and 42.7% N2 in the fuel mixture. Tu = 298 K, p = 1 atm, XN2/XO2 = 3.76. Error bars from [27] are also shown.](/figures/fig-3-laminar-flame-speeds-of-syngas-mixtures-co-h2-ch4-co2-3u6a11oi.png)

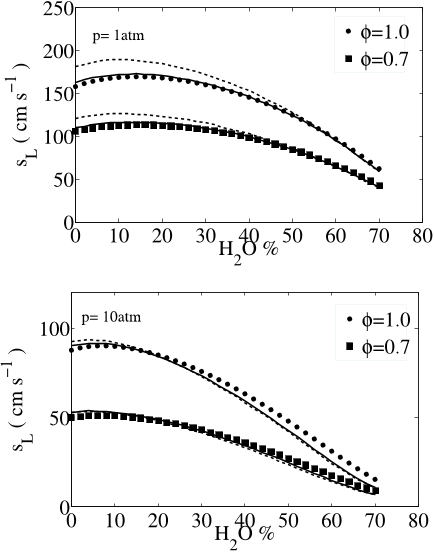
![Fig. 8: Laminar flame speeds of CO/H2 mixtures using the reduced (dashed lines) and skeletal (continuous lines) mechanisms. Symbols: experimental results of Sun et al. [33]. At p = 1 atm the oxidizer is O2,N2 with XN2/XO2 = 3.76. At p = 5, 10, 20 atm the oxidizer is O2 and He with XHe/XO2 = 7.0. Open symbols: experimental results of Singh et al. [21].](/figures/fig-8-laminar-flame-speeds-of-co-h2-mixtures-using-the-1l00yv4j.png)
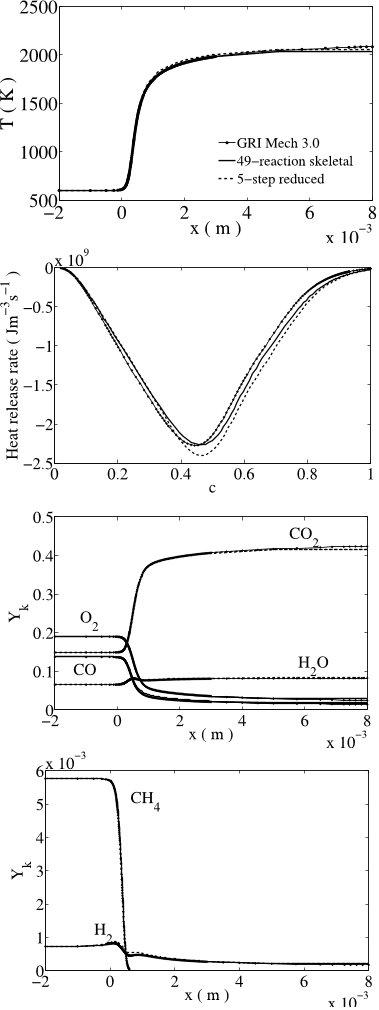
![Fig. 22: Ignition delay times of CO/H2/CO2/O2/N2 mixtures using the reduced (dashed lines) and skeletal (continuous lines) mechanisms. Symbols: experimental data of [28], 8.91%H2 + 11.58%CO+ 24.44%CO2 + 10.25%O2 + 44.83%N2.62](/figures/fig-22-ignition-delay-times-of-co-h2-co2-o2-n2-mixtures-3pfqzjjf.png)
![Fig. 21: Ignition delay times of CO/H2/O2/N2 mixtures ( XN2/XO2 = 3.76) for φ = 0.5 using the reduced (dashed lines) and skeletal (continuous lines) mechanisms. Symbols: experimental results of Kalitan et al. [55]. Also shown for comparison are the results with the skeletal mechanism of Boivin et al. [14] (dashed lines with ×) for the fH2 = 20/80 case.](/figures/fig-21-ignition-delay-times-of-co-h2-o2-n2-mixtures-xn2-xo2-3newhz2i.png)

![Fig. 1: Laminar flame speeds of CO/H2/H2O-air mixtures using the reduced (dashed lines) and skeletal (full lines) mechanisms. Open circles: Li et al. [31] mechanism results from [22]. Also shown are the predictions using the skeletal mechanism of Boivin et al. (dashed lines with ×) [14]. Filled symbols: experimental results of Das et al. [22]. Tu = 323 K, p = 1 atm, fH2 = 5/95, XN2/XO2 = 3.76.](/figures/fig-1-laminar-flame-speeds-of-co-h2-h2o-air-mixtures-using-1wc6l9to.png)
![Fig. 2: Laminar flame speeds of CO/H2/H2O mixtures using the reduced (dashed lines) and skeletal (continuous lines) mechanisms. Filled symbols: experimental results of Singh et al. [21]. p = 1 atm, Tu = 400 K, φ = 1, oxidiser is O2,N2 with XN2/XO2 = 3.76.](/figures/fig-2-laminar-flame-speeds-of-co-h2-h2o-mixtures-using-the-twgf0oeg.png)
![Fig. 7: Laminar flame speeds of CO/H2/CO2/O2/N2 mixtures using the reduced (dashed lines) and skeletal (continuous lines) mechanisms . Symbols: experimental results of Natarajan et al. [49]. fH2 = 5/95 and 1.0, at p = 1 atm, XN2/XO2 = 3.76 with 10% and 20% CO2 dilution.](/figures/fig-7-laminar-flame-speeds-of-co-h2-co2-o2-n2-mixtures-using-3p13osby.png)
![Fig. 6: Laminar flame speeds of CO/H2 mixtures using the reduced (dashed lines) and skeletal (continuous lines) mechanisms. Filled symbols: experimental results of Singh et al. [21]. p = 1 atm, oxidizer is air.](/figures/fig-6-laminar-flame-speeds-of-co-h2-mixtures-using-the-2hspl2i9.png)

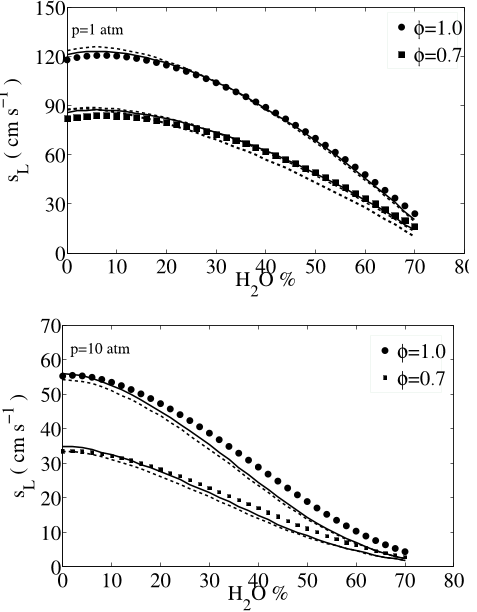


![Fig. 10: Laminar flame speeds of CO/H2/CH4/CO2/O2/N2 mixtures using the reduced (dashed lines) and skeletal (continuous lines) mechanisms. Symbols: experimental results of Park et al. [54], p = 1 atm, Tu = 298 K. 49](/figures/fig-10-laminar-flame-speeds-of-co-h2-ch4-co2-o2-n2-mixtures-1taf19bx.png)

![Fig. 5: Laminar flame speeds of CO/H2/O2/N2 mixtures using the reduced (dashed lines) and skeletal ( continuous lines ) mechanisms. Also shown are the results using the 4- step reduced mechanism of [14] ( open squares ), the skeletal mechanism of [14] ( open circles ) from the same study, and the implementation of the skeletal mechanism of [14] in this study (dashed-dotted lines). Symbols: experimental results of Natarajan et al. [49]. fH2 = 5/95 and 1.0, at p = 1 atm, XN2/XO2 = 3.76, for Tu = 400, 500, 600 and 700 K.](/figures/fig-5-laminar-flame-speeds-of-co-h2-o2-n2-mixtures-using-the-izelzdr2.png)