prognosis or monitoring therapeutic response.
13, 14
The
fundamental goal of developing molecular probes capable of
imaging redox activity has seen some clinical success. In
particular, positron emission tomography (PET) probes capable
of targeting hypoxic tissue, such as the radiotracers
64
Cu
II
-
diacetyl-bis(N4-methylthiosemi-carbazone) (
64
Cu
II
-ATSM) and
18
F-fluoro-misonidazole (
18
F-MISO), showed capacity to predict
treatment outcome in patients undergoing radiotherapy.
15-17
The same hypoxia targeting mechanism of probe uptake
(irreversible reaction and retention in oxygen-deprived tissue)
has been extended to MRI contrast agents and fluorescent
probes for optical imaging.
18, 19
Redox-sensitive nitroxide
derivative probes were also used in electron paramagnetic
resonance (EPR) imaging and spectroscopy to detect reductive
environment (thiols and other reducing species).
20, 21
Magnetic resonance imaging (MRI) is an attractive modality to
monitor redox dynamics thanks to its exceptional spatial
resolution (<100
m using modern high-field equipment), lack
of invasiveness and use of ionizing radiation, and its ability to
simultaneously report physiological and anatomical
information. Not surprisingly, the interest in the development
of redox sensitive MRI probes has grown over the last years.
14,
22-26
These can be based on redox active ligands, undergoing a
change in the ligand structure or the molecular rotation upon
reduction/oxidation, while the oxidation state of the
paramagnetic metal remains the same (e.g. merocyanine unit
linked to a GdDO3A which isomerizes to the spirooxazine
derivative by the redox stimulus of NADH).
27
Alternatively,
complexes of redox active paramagnetic metals, like europium
and manganese, can be also explored, in which the different
oxidation states have different magnetic properties.
11, 22
Utterly, both types of probes must fulfill several requirements:
i) biocompatibility, ii) a redox half-cell potential which is
compatible with the redox potential of biological reducing
agents; iii) strong signal change, if possible signal increase upon
activation (“turn-on” probe); and iv) rapid redox kinetics as
compared to the imaging time scale. Importantly, the redox
active metal ion needs to be complexed by a ligand that is
capable of efficiently chelating both oxidation states such that
reduction or oxidation does not result in the decomposition of
the complex.
Among redox active metal ions, manganese is particularly
attractive for the development of redox responsive MRI probes,
whose efficiency is measured by their proton relaxivity r
i
(i = 1,2)
(paramagnetic enhancement of the water proton relaxation
rates, R
i
= T
i
-1
, where T
1
and T
2
are, respectively, the longitudinal
and transverse relaxation times, referred to 1 mM
concentration of the paramagnetic ion). Manganese is a
biogenic element and in its +2 oxidation state, Mn
2+
, is a very
efficient paramagnetic relaxation agent. Mn
2+
(d
5
configuration,
high spin S = 5/2), with long (in the 0.1 – 1 ns range) longitudinal
electronic relaxation times and labile water exchange, is an
attractive alternative to Gd
3+
in MRI probes.
28, 29
Indeed, in the
last years, there has been intensive research to identify suitable
ligand structures for Mn
2+
chelation.
30, 31
Mn
3+
(d
4
configuration)
is also paramagnetic, usually giving rise to high spin S = 2
compounds. While water exchange is also fast,
14
the
longitudinal electronic relaxation times are shorter (≤ 10 ps),
making them less efficient relaxation agents.
32
The nuclear
relaxation mechanism of Mn
3+
is different from that of Mn
2+
and
therefore their relaxation efficiency shows a very distinct
dependence on the external magnetic field B
0
. While for the S =
5/2 Mn
2+
complexes, the classical Solomon-Bloembergen-
Morgan (SBM) formalism describes well nuclear relaxation,
leading to a decrease of r
1
at high field, for the S = 2 Mn
3+
complexes the analysis of the water proton relaxation rates is
much less straightforward. Water soluble and air stable Mn
3+
-
porphyrins have been investigated as tumor targeted MRI
agents.
33
For these compounds, the electron relaxation times
are relatively long due to their higher symmetry.
32
For instance,
the Mn
III
-TPPS
4
(meso-tetra(4-sulfonatophenyl)porphine
chloride) chelate
34
displays an anomalous r
1
relaxivity, with a
peak above approximately 2 MHz. It was shown that the NMR
relaxation mechanism has several unique aspects, including the
unusual role of large rhombic zero-field splitting (ZFS)
interactions, which are of the order of the nuclear Zeeman
energy. The effect of these interactions on the electron spin
dynamics leads to those relaxivity anomalies at high field.
35,36
As
a consequence of the different relaxation mechanisms, the
proton relaxivity, and thus the MRI efficiency can be either
higher or lower for the reduced Mn
2+
relative to the oxidized
Mn
3+
form, depending on the external magnetic field and on the
chelating ligand.
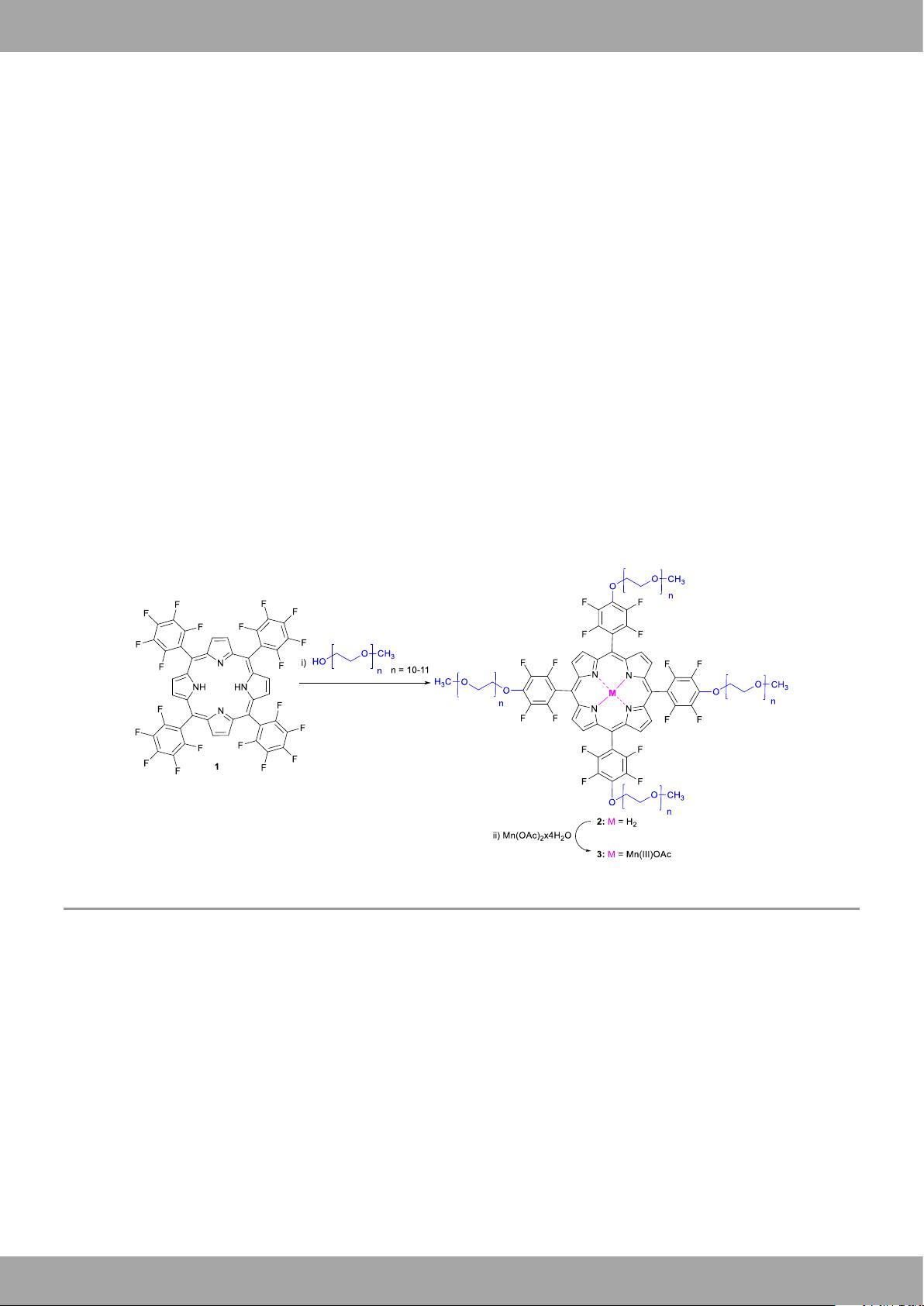
Porphyrins, which are macrocyclic ligands per excellence and
widely used in a multitude of applications, such as catalysis,
37-39
materials science
40-43
and biomedicine, including MRI,
30, 44-48
offer the possibility to obtain very stable chelates to host many
metal ions, including paramagnetic ones. Such
metalloporphyrins having a much reduced risk of
transmetallation, may avoid potential in vivo metal release
during the MRI examination which would cause toxicity, such as
for instance gadolinium induced fibrosis disorders,
49
or
manganese induced neurotoxicity.
50 50 50 50 50 50 50 50 50 50 50
Many
studies have been reported of Mn
III
-porphyrin dimers,
supramolecular and macromolecular conjugates as MRI
contrast agents,
33, 51-58
including enzyme
55, 56
and Zn
2+
-
responsive
57, 58
agents. So far, the only studies on Mn-
porphyrins as redox MRI probes were reported by Aime and
collaborators,
59
who explored a Mn
II
/Mn
III
-based water-soluble
porphyrin (TPPS
4
) as an oxygen sensor. However, at clinically
relevant fields (
1
H frequencies of 60-120 MHz), and at 25 ºC, the
r
1
relaxivity
values revealed to be very close for the Mn
II
and
Mn
III
analogues. On the other hand, the formation of
macromolecular conjugates, through the encapsulation of the
Mn
II
-TPPS
4
and Mn
III
-TPPS
4
complexes in commercially
available, water-soluble poly-
-cyclodextrin (CD), led to a
marked difference in relaxivity at 20 MHz between the reduced
and oxidized state.