
All figures (9)
 Fig. 1. Biosorption capacity of different agro-industrial wastes and by-products.
Fig. 1. Biosorption capacity of different agro-industrial wastes and by-products.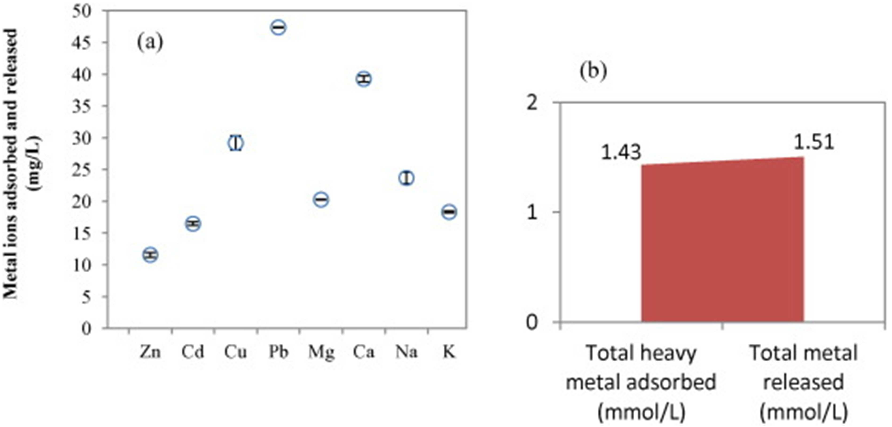 Fig. 4. Comparison of (a) individual and (b) total metal ions adsorbed and released in biosorption process (initial heavy metal conc.: 50 mg/L).
Fig. 4. Comparison of (a) individual and (b) total metal ions adsorbed and released in biosorption process (initial heavy metal conc.: 50 mg/L). Table 4. Biosorption capacities of various adsorbents.
Table 4. Biosorption capacities of various adsorbents.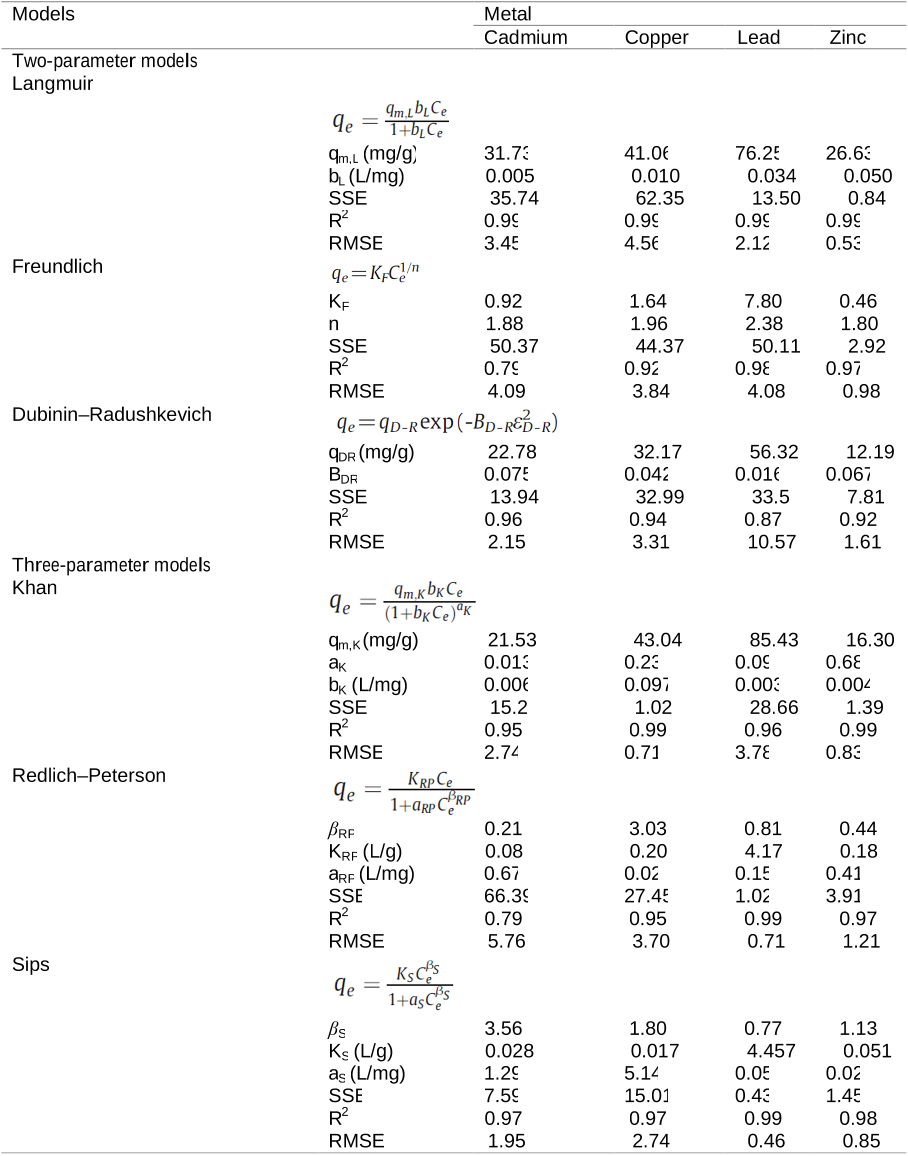 Table 3. Isotherm constants of two- and three-parameter models for Cd(II), Cu(II), Pb(II) and Zn(II) adsorption (initial metal conc.: 10–500 mg/L).
Table 3. Isotherm constants of two- and three-parameter models for Cd(II), Cu(II), Pb(II) and Zn(II) adsorption (initial metal conc.: 10–500 mg/L). Table 1. FTIR spectra and SEM images of (a) unloaded and (b) metal loaded-biosorbents.
Table 1. FTIR spectra and SEM images of (a) unloaded and (b) metal loaded-biosorbents. Fig. 5. Van't Hoff plots for Cd(II), Cu(II), Pb(II) and Zn(II) adsorption(initial pH 5.5 ± 0.1; initial metal conc.: 1–50 mg/L; contact time: 3 h; biosorbent dose: 5 g/L).
Fig. 5. Van't Hoff plots for Cd(II), Cu(II), Pb(II) and Zn(II) adsorption(initial pH 5.5 ± 0.1; initial metal conc.: 1–50 mg/L; contact time: 3 h; biosorbent dose: 5 g/L). Table 2. Adsorption rate constants, the estimated qe and the coefficients of correlation associated with the Lagergren pseudo-first and pseudo-second order, kinetic models.
Table 2. Adsorption rate constants, the estimated qe and the coefficients of correlation associated with the Lagergren pseudo-first and pseudo-second order, kinetic models. Fig. 3. Effect of (a) initial pH of solution, (b) contact time, (c) biosorbent dose and (d) biosorbent particle size on Cd(II), Cu(II), Pb(II) and Zn(II) adsorption (room temperature, 22 ± 2 °C; initial metal conc.: 50 mg/L).
Fig. 3. Effect of (a) initial pH of solution, (b) contact time, (c) biosorbent dose and (d) biosorbent particle size on Cd(II), Cu(II), Pb(II) and Zn(II) adsorption (room temperature, 22 ± 2 °C; initial metal conc.: 50 mg/L).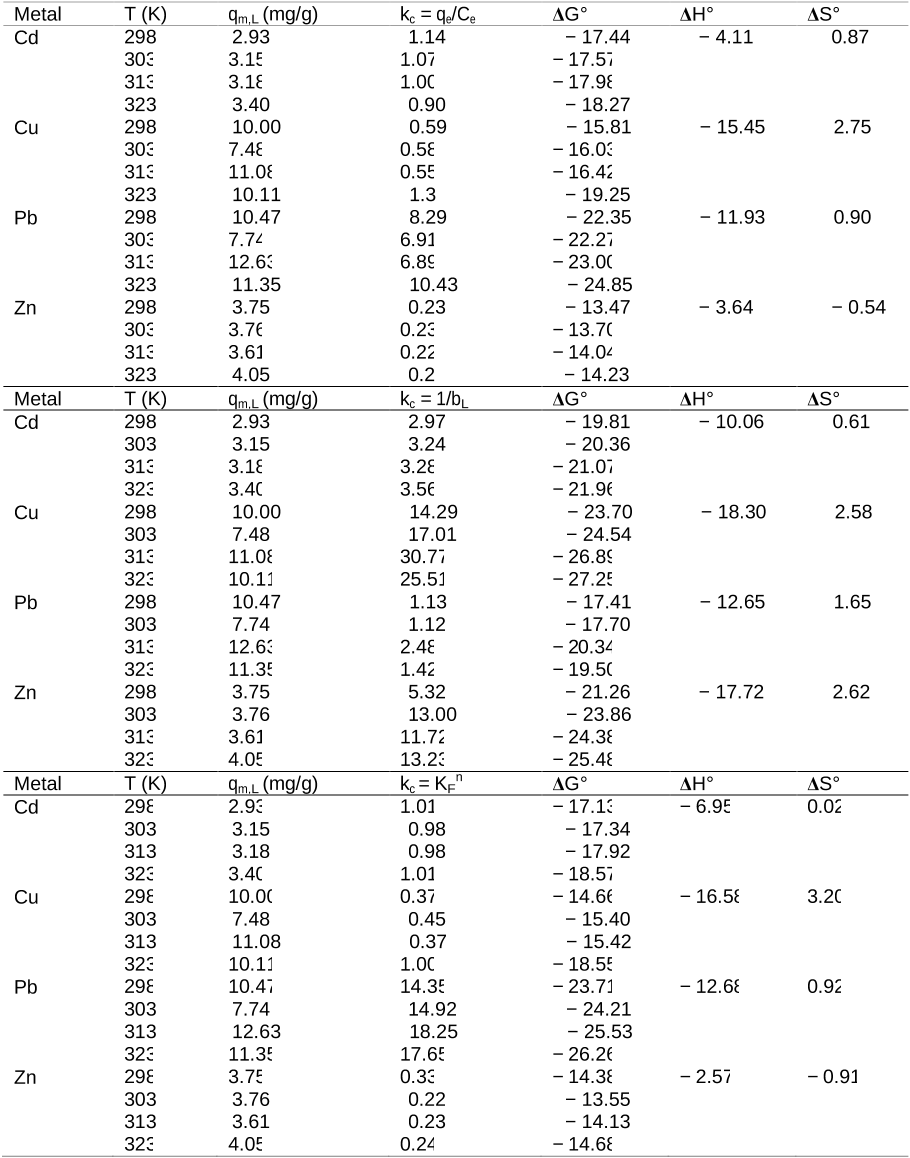 Table 5. Thermodynamic parameters, ∆G° (kJ/mol), ∆H° (kJ/mol) and ∆S° (kJ/mol K), for adsorption of Cd(II), Cu(II), Pb(II) and Zn(II) adsorption.
Table 5. Thermodynamic parameters, ∆G° (kJ/mol), ∆H° (kJ/mol) and ∆S° (kJ/mol K), for adsorption of Cd(II), Cu(II), Pb(II) and Zn(II) adsorption.









