




Did you find this useful? Give us your feedback

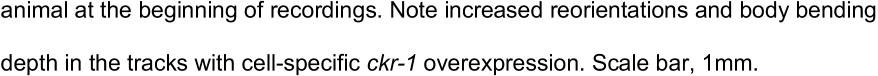





12 citations
10 citations
7 citations
4 citations
219 citations
170 citations
...…after food removal, and decrease with prolonged removal from food (>15-20 minutes) as animals transition to global searching (dispersal) (Bhattacharya et al., 2014; Calhoun et al., 2014; Gray et al., 2005; Hills et al., 2004; Hums et al., 2016; Oranth et al., 2018; Wakabayashi et al., 2004)....
[...]
159 citations
...Brain CCK has been increasingly implicated as a key regulator in diverse aspects of behavior, including feeding, satiety, memory, nociception and anxiety (Ballaz, 2017; Chandra and Liddle, 2007; Liddle, 1997; Miyasaka and Funakoshi, 2003; Noble and Roques, 2006; Rehfeld, 2017)....
[...]
154 citations
...Prior studies of worms immobilized using microfluidic chips and freely moving animals noted anti-phasic activity between SMDD and SMDV neurons and opposing head/neck musculature during head bending (or head casting) (Hendricks et al., 2012; Kaplan et al., 2019; Shen et al., 2016; Yeon et al., 2018)....
[...]
...…regulation of SMD activity is complex and involves reciprocal connections with RIA interneurons, reciprocal signaling with RME motor neurons, as well as proprioceptive feedback (Hendricks et al., 2012; Ouellette et al., 2018; Shen et al., 2016; White, 2018; White et al., 1976; Yeon et al., 2018)....
[...]
...They have been previously implicated in directional head bending and steering (Gray et al., 2005; Hendricks et al., 2012; Kaplan et al., 2019; Kocabas et al., 2012; Shen et al., 2016; Yeon et al., 2018)....
[...]
151 citations
...The lgc-55 promoter drives expression in AVB, RMD, SMD and IL1 neurons, as well as neck muscles and a few other head neurons (Pirri et al., 2009), while the odr-2(16) promoter primarily labels the RME and SMD head neurons (Chou et al., 2001) (Supplementary Files 2-3)....
[...]