
All figures (14)
![Figure 11. ESR spectra recorded for the nickel(III) complex16 and the precipitateP-5 derived from [G1]-Ni12 (5).](/figures/figure-11-esr-spectra-recorded-for-the-nickel-iii-complex16-25cz1zkw.png) Figure 11. ESR spectra recorded for the nickel(III) complex16 and the precipitateP-5 derived from [G1]-Ni12 (5).
Figure 11. ESR spectra recorded for the nickel(III) complex16 and the precipitateP-5 derived from [G1]-Ni12 (5).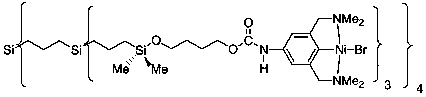 Figure 1. Schematic representation of a metallodendritic catalyst.6
Figure 1. Schematic representation of a metallodendritic catalyst.6 Figure 2. Schematic structure ofpara-functionalized nickel complexes derived from the NCN ligand.
Figure 2. Schematic structure ofpara-functionalized nickel complexes derived from the NCN ligand. Table 1. Cyclic Voltammetry Results of the Model Compounds2 and3 and the Nickelated Dendrimers in CH3CNa,b
Table 1. Cyclic Voltammetry Results of the Model Compounds2 and3 and the Nickelated Dendrimers in CH3CNa,b![Figure 4. Space filling model of the “front” and “side” view of the calculated structure of [G1]-Ni12 (5).](/figures/figure-4-space-filling-model-of-the-front-and-side-view-of-2dtx3nlb.png) Figure 4. Space filling model of the “front” and “side” view of the calculated structure of [G1]-Ni12 (5).
Figure 4. Space filling model of the “front” and “side” view of the calculated structure of [G1]-Ni12 (5).![Figure 3. MALDI-TOF-MS of [G1]-Ni8 (15).](/figures/figure-3-maldi-tof-ms-of-g1-ni8-15-61498krv.png) Figure 3. MALDI-TOF-MS of [G1]-Ni8 (15).
Figure 3. MALDI-TOF-MS of [G1]-Ni8 (15).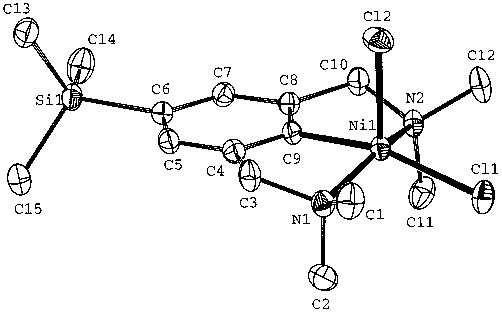 Figure 10. Displacement ellipsoid plot (ORTEP, 50% probability level) of the molecular structure of16. Selected bond lengths (Å) and angles (deg) with esd’s in parentheses: Ni-Cl1 2.2761(5), Ni-Cl2 2.2756(6), Ni-C9 1.8850(16), Ni-N1 2.0324(14), Ni-N2 2.0283(14), Cl1-Ni-C9 164.01(6), N1-Ni-N2 154.05(6), N1-Ni-C9 82.46- (6), Cl1-Ni-N1 93.44(4), Cl2-Ni-N1 100.34(4).
Figure 10. Displacement ellipsoid plot (ORTEP, 50% probability level) of the molecular structure of16. Selected bond lengths (Å) and angles (deg) with esd’s in parentheses: Ni-Cl1 2.2761(5), Ni-Cl2 2.2756(6), Ni-C9 1.8850(16), Ni-N1 2.0324(14), Ni-N2 2.0283(14), Cl1-Ni-C9 164.01(6), N1-Ni-N2 154.05(6), N1-Ni-C9 82.46- (6), Cl1-Ni-N1 93.44(4), Cl2-Ni-N1 100.34(4). Figure 5. Visualization of the catalytic results obtained for nickelated compounds2, 4, 5, and7. Abbreviations used:C ) conversion,t ) time. The curve corresponding to the catalytic performance of3 has been omitted for clarity reasons.
Figure 5. Visualization of the catalytic results obtained for nickelated compounds2, 4, 5, and7. Abbreviations used:C ) conversion,t ) time. The curve corresponding to the catalytic performance of3 has been omitted for clarity reasons. Figure 6. Visualization of the catalytic results obtained for nickelated compounds5, 11, and15. Abbreviations used:C ) conversion,t ) time.
Figure 6. Visualization of the catalytic results obtained for nickelated compounds5, 11, and15. Abbreviations used:C ) conversion,t ) time.![Figure 7. Display of the retained fractions of [G0]-Ni4 (4) and [G1]Ni12 (5) in a membrane reactor.26](/figures/figure-7-display-of-the-retained-fractions-of-g0-ni4-4-and-2buhf0q0.png) Figure 7. Display of the retained fractions of [G0]-Ni4 (4) and [G1]Ni12 (5) in a membrane reactor.26
Figure 7. Display of the retained fractions of [G0]-Ni4 (4) and [G1]Ni12 (5) in a membrane reactor.26![Figure 12. Schematic deactivation mode for dendrimer catalysts [G1]Ni12 (5) and [G2]-Ni36 (7).](/figures/figure-12-schematic-deactivation-mode-for-dendrimer-3vlhomrz.png) Figure 12. Schematic deactivation mode for dendrimer catalysts [G1]Ni12 (5) and [G2]-Ni36 (7).
Figure 12. Schematic deactivation mode for dendrimer catalysts [G1]Ni12 (5) and [G2]-Ni36 (7).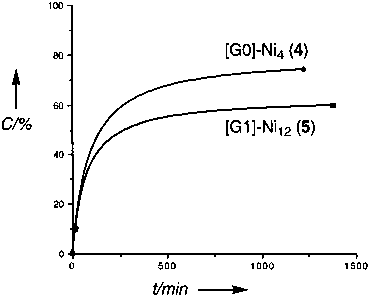 Figure 8. Display of the membrane catalysis with4 and5. Abbreviations used:C ) conversion,t ) time.26
Figure 8. Display of the membrane catalysis with4 and5. Abbreviations used:C ) conversion,t ) time.26 Table 3. Membrane Catalysis Results Obtained for4 and5a
Table 3. Membrane Catalysis Results Obtained for4 and5a Figure 9. Continuous membrane catalysis carried out with5. Abbreviations used: STY) space time yield,t ) time.
Figure 9. Continuous membrane catalysis carried out with5. Abbreviations used: STY) space time yield,t ) time.

![Figure 11. ESR spectra recorded for the nickel(III) complex16 and the precipitateP-5 derived from [G1]-Ni12 (5).](/figures/figure-11-esr-spectra-recorded-for-the-nickel-iii-complex16-25cz1zkw.png)



![Figure 4. Space filling model of the “front” and “side” view of the calculated structure of [G1]-Ni12 (5).](/figures/figure-4-space-filling-model-of-the-front-and-side-view-of-2dtx3nlb.png)
![Figure 3. MALDI-TOF-MS of [G1]-Ni8 (15).](/figures/figure-3-maldi-tof-ms-of-g1-ni8-15-61498krv.png)



![Figure 7. Display of the retained fractions of [G0]-Ni4 (4) and [G1]Ni12 (5) in a membrane reactor.26](/figures/figure-7-display-of-the-retained-fractions-of-g0-ni4-4-and-2buhf0q0.png)
![Figure 12. Schematic deactivation mode for dendrimer catalysts [G1]Ni12 (5) and [G2]-Ni36 (7).](/figures/figure-12-schematic-deactivation-mode-for-dendrimer-3vlhomrz.png)


