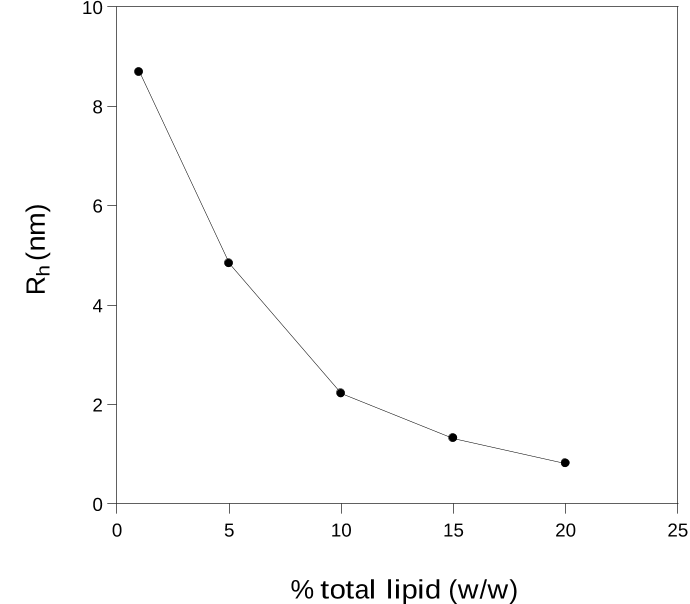
5
specific volume of the 12-carbon lipid tail (Figure 1B). These values can be obtained from the
method described by Durchschlag et al. and in actuality these values vary depending on the
organization and assembly of the lipid aggregate [20, 21]. Although these values are only
estimates, they are sufficiently valid for our study. In the DMPC-DHPC bicelle system, we found
that DHPC imparts a substantial density to the entire bicellar aggregate necessitating the use of
density modifiers. Using the density matching approach in studies of membrane protein
interactions, the contribution of the lipid aggregate to the measured buoyant molecular weight of
an incorporated membrane protein is negated and the oligomeric state of the target membrane
protein can be determined. We sought to investigate the physical properties of a novel lipid
aggregate system with a lower density, yet preserving the bilayer properties of the bicellar
mixtures using DMPC and the detergent, n-octylpentaoxyethylene (C
8
E
5
). C
8
E
5
has a reported
specific volume of 0.993 cm
3
/ g
22
. C
8
E
5
has been used to study membrane protein interactions
in the absence of DMPC in sedimentation equilibrium experiments. With a partial specific volume
(inverse density) substantially similar to water, C
8
E
5
requires little need for density matching
13
.
In the following study, DMPC-C
8
E
5
mixtures were prepared and investigated and their physical
properties compared to DMPC-DHPC bicelles. The size and shape of these lipid-detergent
structures was also investigated using the method of Mazer and co-workers 1980, and later used
by Glover and co-workers to evaluate the structure of DMPC-DHPC bicelles [3,23]. This approach
along with analysis by light scattering was used to predict the shape of DMPC-C
8
E
5
lipid-detergent
aggregates to determine if they assume a discoidal arrangement of lipids and detergent molecules
similar to DMPC-DHPC bicelles. We found that interestingly, these lipid aggregates assume a
shape that is closer to spherical than to discoidal and the properties we observe for DMPC-DHPC
bicelles are not preserved in DMPC-C
8
E
5
aggregates. In and of themselves, DMPC-C
8
E
5
aggregates have very interesting properties and warrant further study for their potential as a
platform in membrane protein analysis and as a vehicle for drug delivery.
.CC-BY-NC-ND 4.0 International licensea
certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under