




Did you find this useful? Give us your feedback










33 citations
33 citations
28 citations
28 citations
24 citations
31,667 citations
25,325 citations
22,053 citations
18,531 citations
17,755 citations
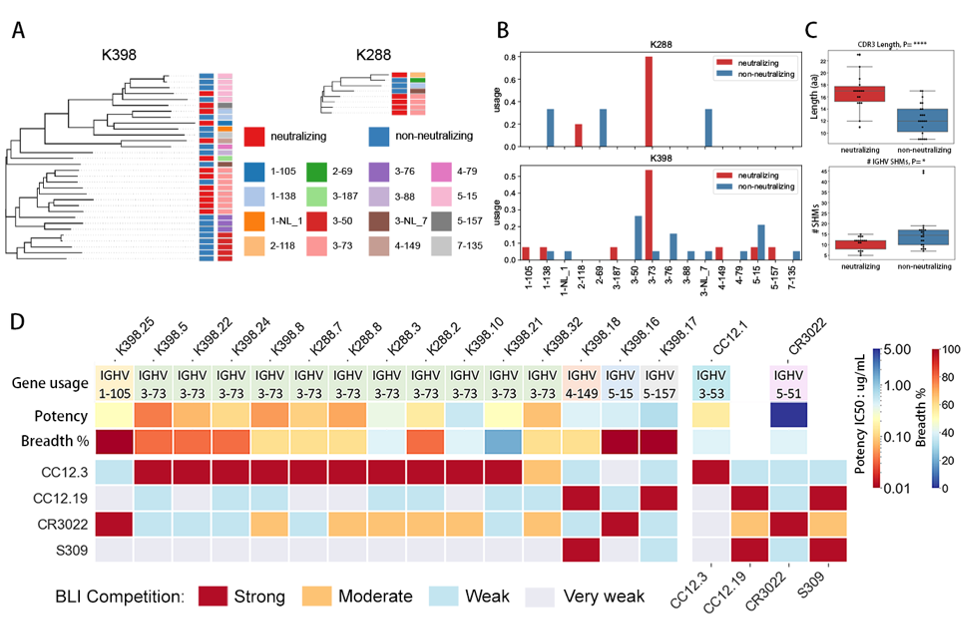
Here, the authors show that immunization of macaques with SARS-CoV-2 spike ( S ) protein generates potent receptor binding domain crossneutralizing antibody ( nAb ) responses to both SARS-CoV-2 and SARS-CoV-1, in contrast to human infection or vaccination where responses are typically SARS-CoV-2-specific. CC-BY-NC-ND 4. 0 International license available under a was not certified by peer review ) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. Furthermore, the macaque nAbs are equally effective against SARS-CoV-2 variants of concern. Human antibodies generally target epitopes strongly overlapping the ACE2 receptor binding site ( RBS ), whereas the macaque antibodies recognize a relatively conserved region proximal to the RBS that represents another potential pan-SARS-related virus site rarely targeted by human antibodies. B cell repertoire differences between the two primates appear to significantly influence the vaccine response and suggest care in the use of rhesus macaques in evaluation of vaccines to SARS-related viruses intended for human use. Broadly neutralizing antibodies to an unappreciated site of conservation in the RBD in SARSrelated viruses can be readily induced in rhesus macaques because of distinct properties of the naïve macaque B cell repertoire that suggest prudence in the use of the macaque model in SARS vaccine evaluation and design.