




Did you find this useful? Give us your feedback























7 citations
6 citations
...The procedure for the CO light-off tests conducted using the SIGU is described by Blades et al.[15] The gas concentrations used were, 10 % CO2, 1 % O2, 0.5 % CO, 5 % H2O and balance N2....
[...]
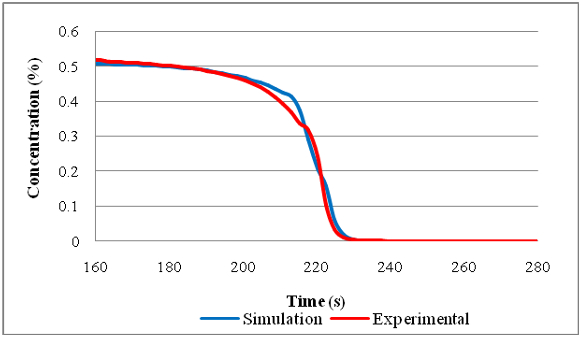
...A study carried out by Blades et al.[15] showed how the QUB global catalyst simulation model, which was first developed by McCullough[16] and later enhanced by Stewart,[17] could accurately simulate the light-off behaviour of catalyst samples for tests conducted on two different laboratory catalyst test rigs....
[...]
...The procedure for the CO light-off tests conducted using the SIGU is described by Blades et al.([15]) The gas concentrations used were, 10 % CO2, 1 % O2, 0....
[...]
...A study carried out by Blades et al.([15]) showed how the QUB global catalyst simulation model, which was first developed by McCullough([16]) and later enhanced by Stewart,([17]) could accurately simulate the light-off behaviour of catalyst samples for tests conducted on two different laboratory catalyst test rigs....
[...]
1 citations
49 citations
...Another form of catalyst deactivation is mechanical deactivation, were the decrease in catalytic activity is caused by loss of catalyst material due to fractures in the ceramic monolith (Zanon Zotin et al., 2005)....
[...]
...…available to reactant gases and therefore reducing the catalyst activity (Harris, 1995; Heck et al., 2002; Martin et al., 2003; Martinez-Arias et al., 2002; Meyer Fernandes et al., 2010; Polvinen et al., 2004; Tanabe et al., 2008; Winkler et al., 2010; Yang et al., 2008; Zanon Zotin et al., 2005)....
[...]
...Research conducted by Zanon Zotin et al. (2005) showed that the deactivation of catalyst samples, aged on an engine bench, was not due to one factor, but a combination of thermal, chemical and mechanical deactivation....
[...]
...Poisoning results in a reduced number of active sites available to the reactant gases and therefore causes reduced catalyst activity (Heck et al., 2002; Zanon Zotin et al., 2005)....
[...]
...…from the gamma phase, γ-Al2O3, through delta, δ-Al2O3 and theta, θ-Al2O3, to the stable alpha alumina, α-Al2O3, with loss of surface area and hence loss of catalyst activity (Heck et al., 2002; Martinez-Arias et al., 2002; Meyer Fernandes et al., 2010; More et al., 1997; Zanon Zotin et al., 2005)....
[...]
45 citations
...At high temperatures, the precious metal particles agglomerate, decreasing the surface area of catalyst available to reactant gases and therefore reducing the catalyst activity (Harris, 1995; Heck et al., 2002; Martin et al., 2003; Martinez-Arias et al., 2002; Meyer Fernandes et al., 2010; Polvinen et al., 2004; Tanabe et al., 2008; Winkler et al., 2010; Yang et al., 2008; Zanon Zotin et al., 2005)....
[...]
...…available to reactant gases and therefore reducing the catalyst activity (Harris, 1995; Heck et al., 2002; Martin et al., 2003; Martinez-Arias et al., 2002; Meyer Fernandes et al., 2010; Polvinen et al., 2004; Tanabe et al., 2008; Winkler et al., 2010; Yang et al., 2008; Zanon Zotin et al., 2005)....
[...]
40 citations
...Moldovan et al. (2003) showed that, after automobile ageing, the loss of precious metal particles, and therefore a reduction in dispersion, was much greater at the front face of a three-way catalyst than in any other region of the brick....
[...]
39 citations
...At high temperatures, the precious metal particles agglomerate, decreasing the surface area of catalyst available to reactant gases and therefore reducing the catalyst activity (Harris, 1995; Heck et al., 2002; Martin et al., 2003; Martinez-Arias et al., 2002; Meyer Fernandes et al., 2010; Polvinen et al., 2004; Tanabe et al., 2008; Winkler et al., 2010; Yang et al., 2008; Zanon Zotin et al., 2005)....
[...]
...The washcoat undergoes irreversible phase changes, with the alumina washcoat transforming from the gamma phase, γ-Al2O3, through delta, δ-Al2O3 and theta, θ-Al2O3, to the stable alpha alumina, α-Al2O3, with loss of surface area and hence loss of catalyst activity (Heck et al., 2002; Martinez-Arias et al., 2002; Meyer Fernandes et al., 2010; More et al., 1997; Zanon Zotin et al., 2005)....
[...]
...A study by Meyer Fernandes et al. (2008) showed that thermal effects were the major contributor to catalyst deactivation....
[...]
...…available to reactant gases and therefore reducing the catalyst activity (Harris, 1995; Heck et al., 2002; Martin et al., 2003; Martinez-Arias et al., 2002; Meyer Fernandes et al., 2010; Polvinen et al., 2004; Tanabe et al., 2008; Winkler et al., 2010; Yang et al., 2008; Zanon Zotin et al., 2005)....
[...]
...…from the gamma phase, γ-Al2O3, through delta, δ-Al2O3 and theta, θ-Al2O3, to the stable alpha alumina, α-Al2O3, with loss of surface area and hence loss of catalyst activity (Heck et al., 2002; Martinez-Arias et al., 2002; Meyer Fernandes et al., 2010; More et al., 1997; Zanon Zotin et al., 2005)....
[...]