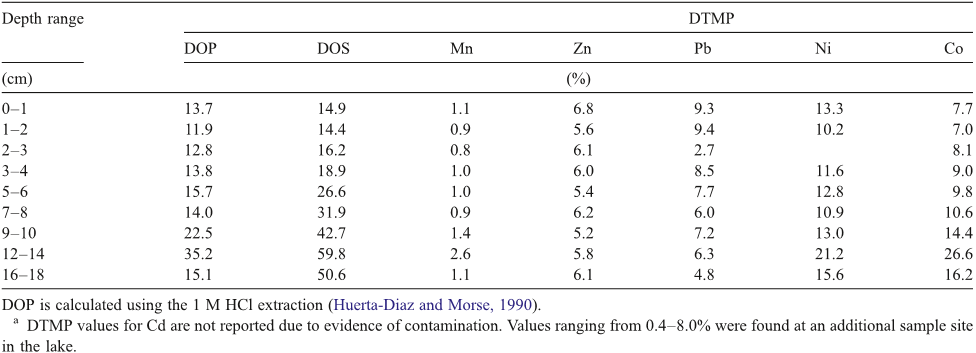
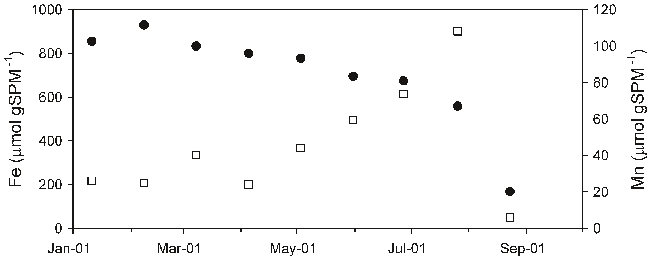
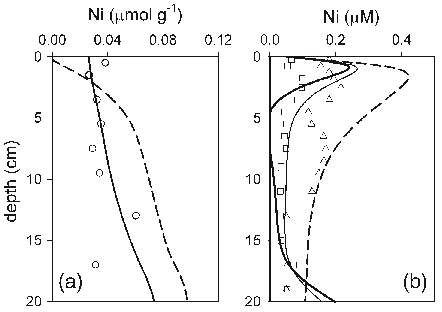
Abstract: The historical trend of heavy metal pollution recorded in sediment cores from Lake Shinji, western Japan, was investigated to evaluate the contribution of increasing long-range transport of heavy metals from the Asian continent in recent years. The concentrations of Cd, Co, Cr, Cu, Ni, Pb, Sb, and Zn and lead isotope ratios were determined for sediment cores collected at two sites in the lake. Among the metals, Cd, Sb, and Zn showed markedly high concentrations since the 1970s. Moreover, a high Pb concentration and less radiogenic lead isotope ratios have been observed since the 1980s in the core from a site close to the mouth of a major river. Air masses from the Asian continent, including China, Russia, and South Korea, have less radiogenic lead isotope ratios than those from Japan. This suggests that the recent increase in Pb concentration in the sediment core is primarily due to the long-range transport of heavy metals from the Asian continent, followed by their deposition in the catchment area of the river. The concentration ratios of Pb/Cd, Pb/Sb, and Pb/Zn of the sediment around 2000 were calculated on the basis of the metal concentrations in excess of those before 1940. They were then compared with the volume-weighted annual average concentration ratios of Pb/Cd, Pb/Sb, and Pb/Zn of rain samples collected on the shore of the lake for 1999–2001. The result showed that the ratios of the former to the latter are 1.0 for Cd, 0.69 for Sb, and 0.31 for Zn. Thus, it is likely that the long-range transport of Cd and Sb from the Asian continent also contributes significantly to the recent increase in the concentrations of these metals in the sediment core from Lake Shinji. For Zn, however, the contribution from the Asian continent was evaluated to be small, suggesting the importance of local sources such as effluent discharges.