




Did you find this useful? Give us your feedback

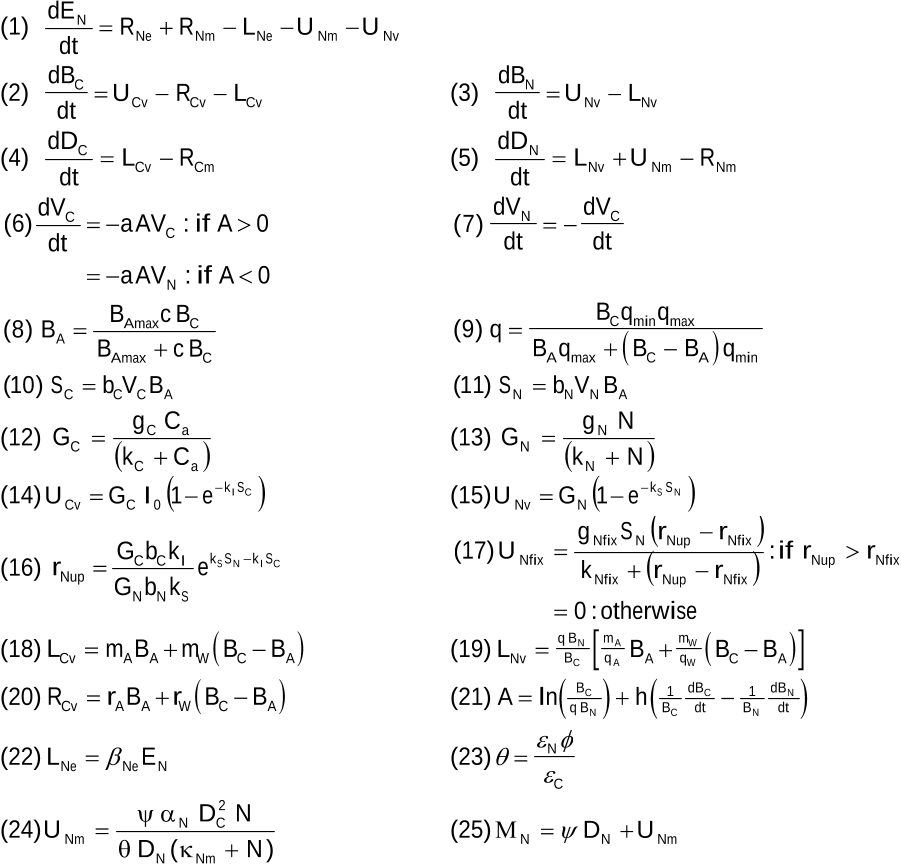





748 citations
...The long-term CO2 stimulation of ecosystem C sequestration on a time scale of decades or longer relies on increases in total ecosystem N stocks (Rastetter et al. 1997, 2005, Luo et al. 2004)....
[...]
358 citations
...This distinction is not reflected in current SOM models of SOM dynamics (Jenkinson et al., 1991; Parton et al. 1991; McGuire et al., 1997; Currie, 2003; Rastetter et al., 2005)....
[...]
321 citations
266 citations
...Some first-ordermodels include nutrients with nonlinear functions that capture the costs and benefits of nutrient acquisition [Rastetter et al., 2001, 2005;Wang et al., 2007; Houlton et al., 2008], but generally apply a first-order approach to soil C decomposition [equation (1)]....
[...]
261 citations
[...]
2,647 citations
2,376 citations
2,126 citations
1,734 citations
1,613 citations
The best potential for testing their ideas in a timely manner would be to experimentally manipulate ecosystems where the masking effects of within-ecosystem responses are likely to be small relative to the effects of DON losses to determine if there is a trend toward high C sequestration with low DON losses relative to DIN losses. Thus the manipulations should be on ecosystems where the C: N ratio of vegetation is low ( i. e., close to the C: N ratio of soils so that the redistribution of N has a smaller effect ), where the vegetation is unlikely to increase in woodiness ( i. e., to avoid the masking effects of increasing C: N ratios ), and where the total throughput of DON plus DIN is high ( i. e., a high potential to sequester N ). Their aim in this paper has been to examine how considering the relative magnitudes of DON versus DIN losses might influence assessments of potential C sequestration in terrestrial ecosystems. Their conclusions are that it is vital to quantify these fluxes at least in regards to evaluations of the long-term potential for C sequestration.
In addition, increases in plant and soil C:N ratios can contribute to the withinecosystem responses and help mask the effects of DON losses.
Terrestrial ecosystems are thought to sequester about 25% of the carbon (C) currentlyemitted through fossil-fuel burning and land-use change (IPCC 2001).
The best potential for testing their ideas in a timely manner would be to experimentally manipulate ecosystems where the masking effects of within-ecosystem responses are likely to be small relative to the effects of DON losses to determine if there is a trend toward high C sequestration with low DON losses relative to DIN losses.
With high DON losses, N gains and losses were small during the first 100 years of all the simulations, and the dynamics in the gradual-change simulations generally lagged behind those in the instantaneous-change simulations by about two decades.
Increases in plant and soil C:N ratios contributed less to C sequestration, but in amounts proportionately equivalent to their contributions in the instantaneous-change simulations.
In this paper the authors argue that the amount of C sequestered in terrestrial ecosystems in response to elevated CO2 depends on the fraction of N losses that are in the form of dissolved organic N (DON) versus dissolved inorganic N (DIN); because plants can curtail DIN losses as N demand increases in response to elevated CO2, but plants have little control over DON losses, the potential for accumulating N by limiting N losses should be small if DON losses are high.
Because the C:N ratio of soils is about 25 and that of plants is about 143 (initial C:N values), this redistribution of N results in a net increase in the amount of C stored per unit N in the ecosystem.
Their assessment of C sequestration in relation to DON losses relies upon threemodifications to what has been called "the standard model" of N accumulation in terrestrial ecosystems (Vitousek et al. 1998).
On average, the ecosystems sequestered only about 1 kg C m-2 between years 60 and 1000 or about 7% of the C sequestered during the first 60 years and 6% of the C sequestered in the ecosystems with low DON losses (Fig. 1).
Sequestration of C continues for the duration of all low-DON-loss simulations, although at a rate that is only about 17% of that during the first 60 years (Fig. 1,Table 3).
Because of the explicit linkages between DOC and DON in the various model structures,simulations with higher DON loss also exhibit higher DOC loss.
with a combination of elevated CO2 and warming, increases in woody tissues and the consequent increase in plant C:N ratio contributed significantly to an increase the C stored per unit N in the ecosystem (Fig. 2).