
All figures (10)
 FIGURE 3—SRXTM images of the megaspore Arcellites sp. (S171531) from the Early Cretaceous Torres Vedras locality, Portugal; dataset acquired using a 203 objective and 20 lm thick LAG:Ce scintillator (voxel size 0.37) at 10 keV (1) and a 403 objective with a 5.9 lm thick LSO:Tb scintillator (voxel size 0.1625) at 10 keV (2–6); images in 1, 2 and 4, 5 have been reconstructed uniquely exploiting absorption contrast and pure edge-enhancement. Images in 3, 6 were obtained using simple phase retrieval (Paganin et al., 2002), but with the subsequent addition of the high frequency component of the original data to minimize loss in spatial resolution inherent in phase retrieval. 1–3, 2-D reconstructions of a transverse section through the body of the megaspore showing wall ultrastructure composed of a thin inner, almost solid, uniform layer, a middle granular-fibrous layer of uneven thickness, and an outer layer penetrated by very fine canals; note difference in resolution between 1 originating from a tomographic volume obtained with a 203objective and a 20 lm thick LAG:Ce screen, and
FIGURE 3—SRXTM images of the megaspore Arcellites sp. (S171531) from the Early Cretaceous Torres Vedras locality, Portugal; dataset acquired using a 203 objective and 20 lm thick LAG:Ce scintillator (voxel size 0.37) at 10 keV (1) and a 403 objective with a 5.9 lm thick LSO:Tb scintillator (voxel size 0.1625) at 10 keV (2–6); images in 1, 2 and 4, 5 have been reconstructed uniquely exploiting absorption contrast and pure edge-enhancement. Images in 3, 6 were obtained using simple phase retrieval (Paganin et al., 2002), but with the subsequent addition of the high frequency component of the original data to minimize loss in spatial resolution inherent in phase retrieval. 1–3, 2-D reconstructions of a transverse section through the body of the megaspore showing wall ultrastructure composed of a thin inner, almost solid, uniform layer, a middle granular-fibrous layer of uneven thickness, and an outer layer penetrated by very fine canals; note difference in resolution between 1 originating from a tomographic volume obtained with a 203objective and a 20 lm thick LAG:Ce screen, and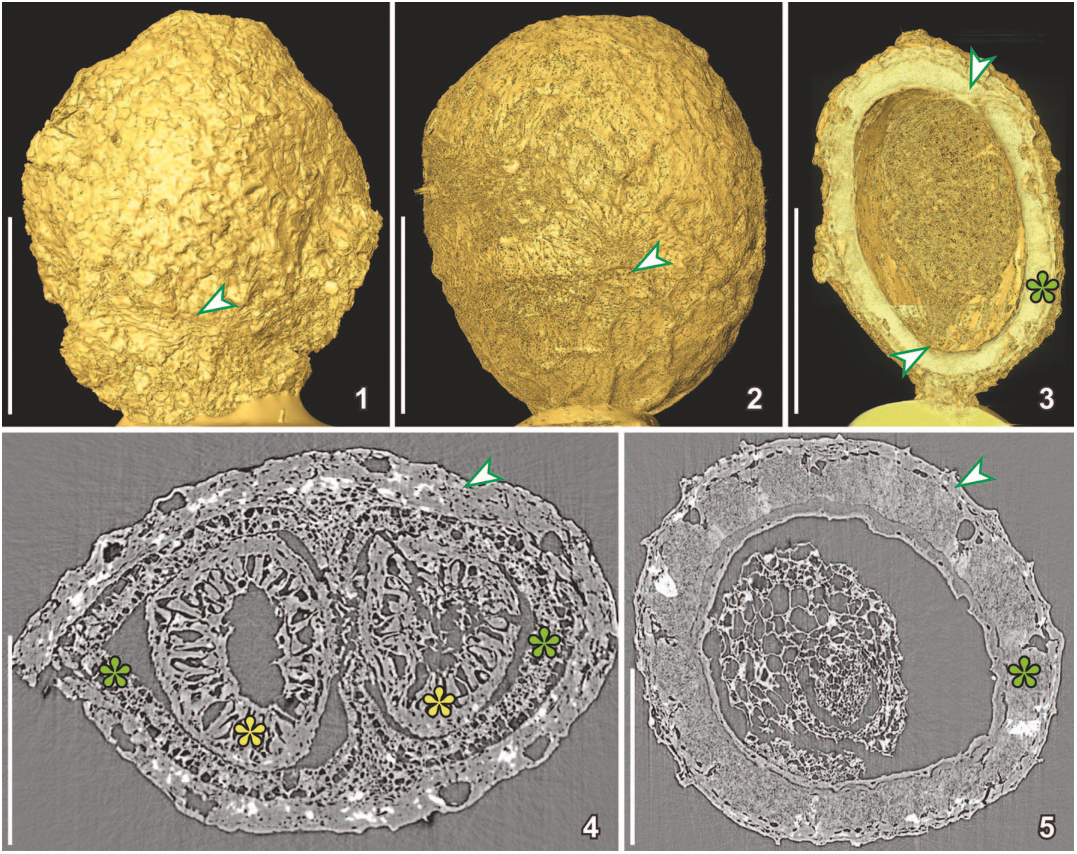 FIGURE 6—Three-dimensional (1–3) and 2-D (4, 5) SRXTM reconstructions of two charcoalified fruits with similar external morphology, but different internal structure; 1, surface rendering of Canrightia resinifera (S171508) from the Early Cretaceous Catefica locality, Portugal, showing slightly wrinkled surface of fleshy fruit wall and short hypanthium (arrowhead); dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 2, surface rendering of Canrightia-like fruit (S174033) from the Early Cretaceous Famalicão locality, Portugal, showing slightly wrinkled surface of fleshy fruit wall and scars from stamens (arrowhead) on the fruit surface; dataset acquired using 203 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.325 lm); 3, combined surface rendering and cut voltex (transparent rendering) of Canrightia-like fruit (S174105) from the Early Cretaceous Buarcos locality, Portugal, in longitudinal section showing a single, hemi-orthotropous and pendulous seed with chalaza and vascular bundles entering the seed close to apex (arrowhead) and micropyle directed towards the base (arrowhead) and with thick crystalliferous endotesta (green asterisk); dataset acquired using 203 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.325 lm); 4, transverse orthoslice of Canrightia resinifera (same dataset as for 1) showing fruit wall (arrowhead) enclosing two ovules/seeds with the seed wall composed of crystalliferous and fibrous endotesta (green asterisks) and a thick endotegmen composed mainly of endothelium cells (yellow asterisks); 5, transverse orthoslice of Canrightia-like fruit with collapsed fruit wall (arrowhead) enclosing a single seed (same dataset as for 2); seed wall mainly composed of crystalliferous and fibrous endotesta and with grooves giving the seed surface a pitted appearance; inside are remains of endosperm and embryo. Scale bars for 1–3¼500 lm; for 4, 5¼250 lm.
FIGURE 6—Three-dimensional (1–3) and 2-D (4, 5) SRXTM reconstructions of two charcoalified fruits with similar external morphology, but different internal structure; 1, surface rendering of Canrightia resinifera (S171508) from the Early Cretaceous Catefica locality, Portugal, showing slightly wrinkled surface of fleshy fruit wall and short hypanthium (arrowhead); dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 2, surface rendering of Canrightia-like fruit (S174033) from the Early Cretaceous Famalicão locality, Portugal, showing slightly wrinkled surface of fleshy fruit wall and scars from stamens (arrowhead) on the fruit surface; dataset acquired using 203 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.325 lm); 3, combined surface rendering and cut voltex (transparent rendering) of Canrightia-like fruit (S174105) from the Early Cretaceous Buarcos locality, Portugal, in longitudinal section showing a single, hemi-orthotropous and pendulous seed with chalaza and vascular bundles entering the seed close to apex (arrowhead) and micropyle directed towards the base (arrowhead) and with thick crystalliferous endotesta (green asterisk); dataset acquired using 203 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.325 lm); 4, transverse orthoslice of Canrightia resinifera (same dataset as for 1) showing fruit wall (arrowhead) enclosing two ovules/seeds with the seed wall composed of crystalliferous and fibrous endotesta (green asterisks) and a thick endotegmen composed mainly of endothelium cells (yellow asterisks); 5, transverse orthoslice of Canrightia-like fruit with collapsed fruit wall (arrowhead) enclosing a single seed (same dataset as for 2); seed wall mainly composed of crystalliferous and fibrous endotesta and with grooves giving the seed surface a pitted appearance; inside are remains of endosperm and embryo. Scale bars for 1–3¼500 lm; for 4, 5¼250 lm. Figure 9—1–3, SRXTM reconstructions of a flower of extant Quintinia quatrefagesii from North Queensland, Australia, dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm) with vertical stacking; specimen critically point dried prior to SRXTM; 1, 3-D cut voltex (transparent rendering) reconstruction of flower bud in lateral view showing internal details of flower and densely-spaced crystals, seen as whitish flecks, particularly in the anthers (yellow asterisks) and carpels (blue asterisks); 2, 3-D voltex (transparent rendering) reconstruction of flower bud in apical view showing the pentamerous organization with the five stamens (yellow asterisks) and three styles (blue asterisks) demarcated by concentrations of crystals; 3, 2-D transverse orthoslice in the region of the styles with dorsal palisade cells (blue asterisk) of the styles and postgenital fusion of the carpels (arrowhead); 4–6, SRXTM reconstructions of Silvianthemum suecicum from the Late Cretaceous Åsen locality, Sweden; 4, 5, 3-D cut voltex (transparent rendering) reconstruction of post-anthetic flower (S171578) in longitudinal (4) and transverse (5) orientations showing prominent palisade cells of the styles (blue asterisks) and placentation that extends into the apocarpous region of the carpels; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 6, 2-D transverse orthoslice of a flower bud (holotype of Silvianthemum suecicum) through the three styles, showing palisade cells (blue asterisk) of styles extending from ventral to dorsal side and postgenital fusion of carpels (arrowhead) (same dataset as for Fig. 2). Scale bars for 1, 2, 4, 5¼500 lm; for 3, 6¼250 lm.
Figure 9—1–3, SRXTM reconstructions of a flower of extant Quintinia quatrefagesii from North Queensland, Australia, dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm) with vertical stacking; specimen critically point dried prior to SRXTM; 1, 3-D cut voltex (transparent rendering) reconstruction of flower bud in lateral view showing internal details of flower and densely-spaced crystals, seen as whitish flecks, particularly in the anthers (yellow asterisks) and carpels (blue asterisks); 2, 3-D voltex (transparent rendering) reconstruction of flower bud in apical view showing the pentamerous organization with the five stamens (yellow asterisks) and three styles (blue asterisks) demarcated by concentrations of crystals; 3, 2-D transverse orthoslice in the region of the styles with dorsal palisade cells (blue asterisk) of the styles and postgenital fusion of the carpels (arrowhead); 4–6, SRXTM reconstructions of Silvianthemum suecicum from the Late Cretaceous Åsen locality, Sweden; 4, 5, 3-D cut voltex (transparent rendering) reconstruction of post-anthetic flower (S171578) in longitudinal (4) and transverse (5) orientations showing prominent palisade cells of the styles (blue asterisks) and placentation that extends into the apocarpous region of the carpels; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 6, 2-D transverse orthoslice of a flower bud (holotype of Silvianthemum suecicum) through the three styles, showing palisade cells (blue asterisk) of styles extending from ventral to dorsal side and postgenital fusion of carpels (arrowhead) (same dataset as for Fig. 2). Scale bars for 1, 2, 4, 5¼500 lm; for 3, 6¼250 lm. FIGURE 5—Three-dimensional (1, 2, 4–6) and 2-D (3) SRXTM reconstructions of the lignitized flower Monetianthus mirus (S122015) from the Early Cretaceous Vale de Agua locality, Portugal; dataset acquired using 43 objective and a 20 lm thick YAG:Ce scintillator at 10 keV (voltex size 1.85 lm); specimen gold coated and studied by SEM prior to the SRXTM analysis; manipulation with Avizo software highlight the gold cover that has higher density than the coalified tissue; 1, 2, 3-D surface rendering of fossil in two different lateral views showing scars from perianth and androecium; 3, 3-D surface rendering of selected ovules superimposed on an 2-D orthoslice showing anatropous ovules with thin funicles directed towards the septum (laminar placentation); 4–6, 3-D reconstructions combining cut voltex (transparent rendering) and surface rendering of fossil in two different lateral views (4, 5) and in apical view (6) showing syncarpous ovary with many small ovules that do not fill the ovary cavity, section in 4 between orthoslices yz380 and 320, section in 5 between orthoslices xz720–750 and section in 6 between xy620–670. Scale bars for 1, 2, 4–6¼500 lm; for 3¼250 lm.
FIGURE 5—Three-dimensional (1, 2, 4–6) and 2-D (3) SRXTM reconstructions of the lignitized flower Monetianthus mirus (S122015) from the Early Cretaceous Vale de Agua locality, Portugal; dataset acquired using 43 objective and a 20 lm thick YAG:Ce scintillator at 10 keV (voltex size 1.85 lm); specimen gold coated and studied by SEM prior to the SRXTM analysis; manipulation with Avizo software highlight the gold cover that has higher density than the coalified tissue; 1, 2, 3-D surface rendering of fossil in two different lateral views showing scars from perianth and androecium; 3, 3-D surface rendering of selected ovules superimposed on an 2-D orthoslice showing anatropous ovules with thin funicles directed towards the septum (laminar placentation); 4–6, 3-D reconstructions combining cut voltex (transparent rendering) and surface rendering of fossil in two different lateral views (4, 5) and in apical view (6) showing syncarpous ovary with many small ovules that do not fill the ovary cavity, section in 4 between orthoslices yz380 and 320, section in 5 between orthoslices xz720–750 and section in 6 between xy620–670. Scale bars for 1, 2, 4–6¼500 lm; for 3¼250 lm. Figure 8—SRXTM reconstructions of Scandianthus costatus from the Late Cretaceous Åsen locality, Sweden; 1–3, 3-D cut longitudinal voltex reconstructions (transparent renderings) of small lignitized flower bud (S172368) showing inferior ovary and sepals and petals covering androecium and styles (1, 2), inner whorl of stamens closely adpressed to the two free styles (2), and stamens with short filaments and sagittate, dorsifixed anthers (1–3); section in 1 between orthoslices yz875 and 1075, section in 2 between orthoslices yz770–870 and section in 3 between yz440–560; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 4–9, reconstructions of mature, charcoalified specimens (S174119) with strongly ribbed inferior ovary and persistent calyx; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.65 lm); 4–6, 3-D reconstructions of flower in cut longitudinal voltex (transparent rendering) (4) and cut isosurface rendering (5, 6), showing persistent sepals (4, 5), stamens with extended filaments, but
Figure 8—SRXTM reconstructions of Scandianthus costatus from the Late Cretaceous Åsen locality, Sweden; 1–3, 3-D cut longitudinal voltex reconstructions (transparent renderings) of small lignitized flower bud (S172368) showing inferior ovary and sepals and petals covering androecium and styles (1, 2), inner whorl of stamens closely adpressed to the two free styles (2), and stamens with short filaments and sagittate, dorsifixed anthers (1–3); section in 1 between orthoslices yz875 and 1075, section in 2 between orthoslices yz770–870 and section in 3 between yz440–560; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 4–9, reconstructions of mature, charcoalified specimens (S174119) with strongly ribbed inferior ovary and persistent calyx; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.65 lm); 4–6, 3-D reconstructions of flower in cut longitudinal voltex (transparent rendering) (4) and cut isosurface rendering (5, 6), showing persistent sepals (4, 5), stamens with extended filaments, but FIGURE 1—Close-up view of the TOMCAT end station showing the sample holder with a fossil flower mounted on a brass stub, 3 mm in diameter, in front of the microscope. (Paul Scherrer Institute photo).
FIGURE 1—Close-up view of the TOMCAT end station showing the sample holder with a fossil flower mounted on a brass stub, 3 mm in diameter, in front of the microscope. (Paul Scherrer Institute photo). FIGURE 4—SRXTM reconstructions (1–3, 5–7) and SEM image (4) of charcoalified fossil flowers from the Early (1–3) and Late (4–7) Cretaceous of Portugal showing volume rendering and voltex reconstructions of floral parts; 1–3, fragments of unnamed pentamerous fossil flower (S171526) from the Early Cretaceous Catefica locality, Portugal, showing two bulky tepals, each almost completely enveloping a stamen; the thick transparent voltex section (1) and the surface renderings embedded in the voltex reconstruction (2, 3) show the extensive development of the connective between the small pollen sacs and gradual transition from the bulky filament into the basifixed anther; dataset acquired using 203objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.37); 4–7, unnamed primuloid flower from the Late Cretaceous Mira locality, Portugal; 4, external morphology of flower (S170155); 5, 3-D SRXTM reconstruction of same flower in 4; cut voltex section (transparent rendering between orthoslices 570–660) showing the mushroom-shaped placenta in longitudinal section bearing tiny, densely crowded ovules; pyrite infilling in the tissues is seen as lighter yellow regions; dataset acquired using 103objective and a 20 lm thick LAG:Ce scintillator at 12 keV (voxel size 0.74); 6, 7, 3-D SRXTM reconstruction of placenta region of flower (S153146) in lateral (6) and apical (7) view superimposed on phase retrieved orthoslices; dataset acquired using 103 objective and a 20 lm thick YAG:Ce scintillator at 10 keV (voxel size 0.74 lm). Scale bars for 1–5¼500 lm; for 6, 7¼150 lm.
FIGURE 4—SRXTM reconstructions (1–3, 5–7) and SEM image (4) of charcoalified fossil flowers from the Early (1–3) and Late (4–7) Cretaceous of Portugal showing volume rendering and voltex reconstructions of floral parts; 1–3, fragments of unnamed pentamerous fossil flower (S171526) from the Early Cretaceous Catefica locality, Portugal, showing two bulky tepals, each almost completely enveloping a stamen; the thick transparent voltex section (1) and the surface renderings embedded in the voltex reconstruction (2, 3) show the extensive development of the connective between the small pollen sacs and gradual transition from the bulky filament into the basifixed anther; dataset acquired using 203objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.37); 4–7, unnamed primuloid flower from the Late Cretaceous Mira locality, Portugal; 4, external morphology of flower (S170155); 5, 3-D SRXTM reconstruction of same flower in 4; cut voltex section (transparent rendering between orthoslices 570–660) showing the mushroom-shaped placenta in longitudinal section bearing tiny, densely crowded ovules; pyrite infilling in the tissues is seen as lighter yellow regions; dataset acquired using 103objective and a 20 lm thick LAG:Ce scintillator at 12 keV (voxel size 0.74); 6, 7, 3-D SRXTM reconstruction of placenta region of flower (S153146) in lateral (6) and apical (7) view superimposed on phase retrieved orthoslices; dataset acquired using 103 objective and a 20 lm thick YAG:Ce scintillator at 10 keV (voxel size 0.74 lm). Scale bars for 1–5¼500 lm; for 6, 7¼150 lm. FIGURE 2—SRXTM images of holotype of Silvianthemum suecicum (S100376) from the Late Cretaceous Åsen locality, Sweden; dataset acquired using a 103 objective and 20 lm thick LAG:Ce scintillator (voxel size 0.74) at 10 keV; specimen charcoalified and mounted obliquely on SEM stub. 1, 2, 2-D dark and flat field corrected radiographic projections for two different sample orientations; 3, 2-D orthoslice of flower bud in longitudinal view through one style, sample not re-oriented; 4, 5, 3-D surface rendering showing external morphology of flower bud in apical and lateral views, respectively; 6, electronically re-oriented longitudinal section through center of flower bud in 3-D cut voltex (transparent rendering between orthoslices 740–780). Scale bars¼500 lm.
FIGURE 2—SRXTM images of holotype of Silvianthemum suecicum (S100376) from the Late Cretaceous Åsen locality, Sweden; dataset acquired using a 103 objective and 20 lm thick LAG:Ce scintillator (voxel size 0.74) at 10 keV; specimen charcoalified and mounted obliquely on SEM stub. 1, 2, 2-D dark and flat field corrected radiographic projections for two different sample orientations; 3, 2-D orthoslice of flower bud in longitudinal view through one style, sample not re-oriented; 4, 5, 3-D surface rendering showing external morphology of flower bud in apical and lateral views, respectively; 6, electronically re-oriented longitudinal section through center of flower bud in 3-D cut voltex (transparent rendering between orthoslices 740–780). Scale bars¼500 lm. FIGURE 7—SEM image (1), 3-D (2, 3), and 2-D (4–6) SRXTM reconstructions of two charcoalified flower buds of Scandianthus costatus (S172382) from the Late Cretaceous Åsen locality, Sweden; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 1, morphology of flower buds showing slightly worn surface; 2, SRXTM volume rendering showing flower buds and supporting bract (viewed from the opposite direction to that in 1); 3, voltex reconstruction (transparent rendering) of same view as in 1; 4, longitudinal orthoslice through central part of flowers showing apical placentae (arrowheads) in left flower; 5, transverse orthoslice showing, from the outside toward the center, remains of three sepals, a whorl of five bulky petals (one marked with green asterisk), two whorls of five stamens (one marked with yellow asterisk), each stamen with two pairs of pollen sacs, and in the center two free styles (arrowhead); 6, transverse orthoslice through the ovary showing the two apical placentae (arrowheads); ovules are not developed in this very young flower bud. Scale bars for 1–4¼500 lm; for 5, 6¼250 lm.
FIGURE 7—SEM image (1), 3-D (2, 3), and 2-D (4–6) SRXTM reconstructions of two charcoalified flower buds of Scandianthus costatus (S172382) from the Late Cretaceous Åsen locality, Sweden; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 1, morphology of flower buds showing slightly worn surface; 2, SRXTM volume rendering showing flower buds and supporting bract (viewed from the opposite direction to that in 1); 3, voltex reconstruction (transparent rendering) of same view as in 1; 4, longitudinal orthoslice through central part of flowers showing apical placentae (arrowheads) in left flower; 5, transverse orthoslice showing, from the outside toward the center, remains of three sepals, a whorl of five bulky petals (one marked with green asterisk), two whorls of five stamens (one marked with yellow asterisk), each stamen with two pairs of pollen sacs, and in the center two free styles (arrowhead); 6, transverse orthoslice through the ovary showing the two apical placentae (arrowheads); ovules are not developed in this very young flower bud. Scale bars for 1–4¼500 lm; for 5, 6¼250 lm. Figure 10—SRXTM reconstructions of chlamydospermous seeds from the Early Cretaceous of North America (1, 3, 4–6) and Portugal (2). 1, 3-D voltex (transparent rendering) reconstruction of seed of Acanthocatia virginiensis Friis, Pedersen and Crane, 2013a (PP53723), from the Puddledock locality in lateral view showing the rugulate surface and the shape of the sclerenchyma cells; dataset acquired using 203objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.37 lm); 2, 3-D voltex (transparent rendering) reconstruction of seed of Tomcatia taylorii Friis, Pedersen and Crane, 2013a (S154562), from the Torres Vedras locality, in lateral view showing the distinct apical projections (arrowhead) formed from tubular cells that are partly filled by crystals, seen as lighter flecks; dataset acquired using 203 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.37 lm); 3, 3-D voltex (transparent rendering) reconstruction of seed of Cattomia trapezoides Friis, Pedersen and Crane, 2013a (PP53720), from the Puddledock locality in lateral view showing the distinct basal skirt (arrowhead) comprised of tubular cells; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 4–6, 3-D cut voltex (transparent rendering) reconstruction of Acanthocatia virginiensis (same dataset as for 1); 4, longitudinal section through the apical part of the seed showing thick outer seed envelope of sclerenchyma cells enclosing the membranous integument that extends apically into long micropylar tube (yellow asterisk) and papillate lining of sclerenchyma in the micropylar region (arrow); 5, transverse section through the seed in micropylar region showing the micropylar canal closed by radiating cells of inner epidermis of integument (arrowhead) and papillate lining of the sclerenchyma layer (arrow); 6, transverse section through the middle of seed showing the thick sclerenchyma layer of the seed envelope with thin-walled inner epidermal cell (arrow) surrounding the thin tissues of integument and nucellus. Scale bars for 1–3¼500 lm; for 4–6¼250 lm.
Figure 10—SRXTM reconstructions of chlamydospermous seeds from the Early Cretaceous of North America (1, 3, 4–6) and Portugal (2). 1, 3-D voltex (transparent rendering) reconstruction of seed of Acanthocatia virginiensis Friis, Pedersen and Crane, 2013a (PP53723), from the Puddledock locality in lateral view showing the rugulate surface and the shape of the sclerenchyma cells; dataset acquired using 203objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.37 lm); 2, 3-D voltex (transparent rendering) reconstruction of seed of Tomcatia taylorii Friis, Pedersen and Crane, 2013a (S154562), from the Torres Vedras locality, in lateral view showing the distinct apical projections (arrowhead) formed from tubular cells that are partly filled by crystals, seen as lighter flecks; dataset acquired using 203 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.37 lm); 3, 3-D voltex (transparent rendering) reconstruction of seed of Cattomia trapezoides Friis, Pedersen and Crane, 2013a (PP53720), from the Puddledock locality in lateral view showing the distinct basal skirt (arrowhead) comprised of tubular cells; dataset acquired using 103 objective and a 20 lm thick LAG:Ce scintillator at 10 keV (voxel size 0.74 lm); 4–6, 3-D cut voltex (transparent rendering) reconstruction of Acanthocatia virginiensis (same dataset as for 1); 4, longitudinal section through the apical part of the seed showing thick outer seed envelope of sclerenchyma cells enclosing the membranous integument that extends apically into long micropylar tube (yellow asterisk) and papillate lining of sclerenchyma in the micropylar region (arrow); 5, transverse section through the seed in micropylar region showing the micropylar canal closed by radiating cells of inner epidermis of integument (arrowhead) and papillate lining of the sclerenchyma layer (arrow); 6, transverse section through the middle of seed showing the thick sclerenchyma layer of the seed envelope with thin-walled inner epidermal cell (arrow) surrounding the thin tissues of integument and nucellus. Scale bars for 1–3¼500 lm; for 4–6¼250 lm.










