




read more










In this paper, the authors evaluated the performance of two prototype hydrogen fuel storage technologies and concluded that neither was ready to proceed to the commercialization phase of product development without further research.
The data collected on the two prototype hydrogen fuel storage technologies was adequate to determine that neither was ready to proceed to the commercialization phase of product development without further research. While the level of performance achieved did not reach the level needed to move to the commercialization phase of product development, several promising conclusions were reached, resulting in recommendations for future research. Recommendation – Further studies of catalysts should be performed to determine if a higher rate and higher reaction completion can be achieved. 9 % propylamine reacted, but only 9. 0 % propionitrile produced ; the remainder was unwanted byproducts, including some potentially hazardous byproducts, such as cyanide gas.
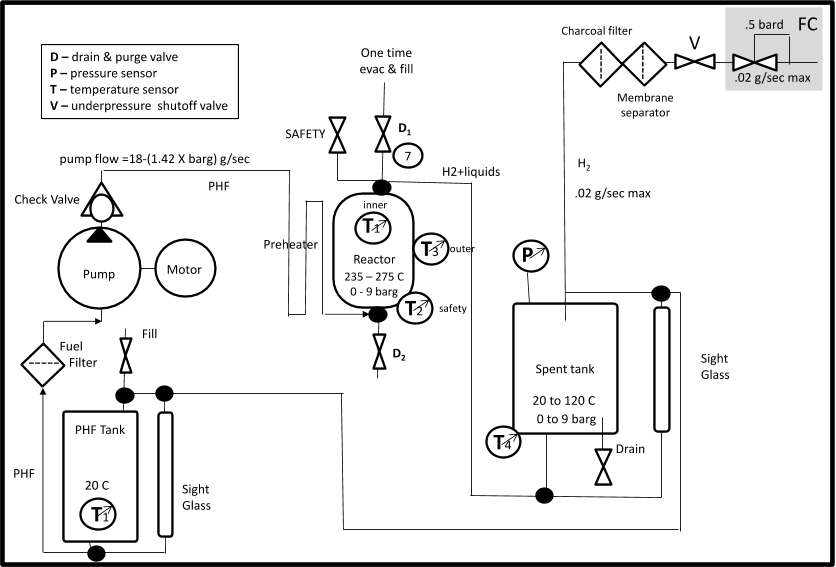
Prior to building the demonstration fuel system, a small, experimental reactor was constructed to aid in selecting the best catalyst and to optimize the reaction conditions.
(16) AP found that catalyst efficiency degraded with reactor length, possibly due to the large volume of hydrogen gas produced (approximately 98% by volume).
Liquid carriers can be stored in conventional fuel tanks that can be conformally mounted, thus simplifying their integration into the vehicle.
The first step toward building the Hydrnol fuel system was to select the reactor components, in particular the catalyst, and collect reactor performance data that would allow accurate sizing of the prototype system.
The dehydrogenation of propylamine to propionitrile was repeated under the same reaction conditions, using the second catalyst, HDS-2A.
Entrepreneurs are especially active at the interface among these traditional business models because the most attractive opportunities to accelerate the pace of change reside there.
neat fuel was chosen for use in the demonstration reactor, and the reactor was designed to provide increased residence time to offset the lower reaction rate of the neat fuel.
Consider BYD, for example, the Chinese battery manufacturer that is integrating into electric vehicles with investment from Warren Buffett.
In addition, this fuel system could be used as the hydrogen source for larger engines that use a small quantity of hydrogen to enhance the combustion process.
a saturated solution of fluorene was desired for the hydrogenation reactions in order to maximize the amount of fluorene hydrogenated.
It was hypothesized that this large volume of gas might form a gas film on the catalyst surface, preventing the liquid from making contact and inhibiting further reaction.
These traditional business models (illustrated in Figure 7) include: Auto companies, once vertically integrated, now requiring innovation by suppliers at all tiers, but still lacking systematic connections with entrepreneurs and new ventures; Energy companies, traditionally commodity energy providers, but increasingly facing competition from “smart” electricity and renewable electricity generated from distributed sources; and Information and communication companies, offering smart devices and systems that manage electric energy onboard vehicles, among energy-using devices, and within smart grids.
Using the HDS-20A catalyst, the calculated percent conversion of propylamine to propionitrile from two duplicate test runs was extremely low even after the reaction temperature was increased from 513K to 573K, as shown in Tables 2 and 3, despite the endothermicity of the dehydrogenation reaction.
The process flow diagram, shown in Figure 12, depicts how the flow of the nitrogen-propylamine mixture was regulated through the use of mass flow controllers and three four-way valves.