




read more









Pore water profiles can be used to assess microbial activity within sediments and the diffusive exchange between sediments and the overlying water.
The diffusive penetration of oxygen into surface sediments, which depends on the oxygen concentration of the overlying water and the oxygen consumption rate in the sediment (mainly aerobic mineralization of organic matter and oxidation of reduced sulphur), determines the sediment depth where sulphate reduction can principally proceed from a thermodynamic viewpoint.
Besides groundwater inflow, post-depositional ion release from this debris, in particular release of mono- and divalent cations and silica may significantly contribute to the DSC of the lake water.
Presuming that LGM and LPM are fed by groundwater inflow of similar composition, an inhibited P-cycle in LPM due to meromixis should be the major reason for the overall higher Si consumption in the lake water of LGM.
Sulphate is utilized as an electron acceptor for the bio-degradation of organic matter in the absence of other preferentially used electron acceptors (O2, NO3, reactive Fe(III), e.g. Furrer and Wehrli 1996) and is consumed by anaerobic methane oxidation (e.g. Iversen and Jørgensen 1985).
Co-precipitation with FeOOH is an important mechanism that can transfer dissolved oxy-anions of U(VI) and Mo(VI) from the water column into the sediments (e.g. Bruno et al. 1995; Gustafsson 2003).
In the nearly non-calcareous sediments from deeper parts of the lake basin, seasonal production of alkalinity associated with sulphate reduction is important.
Elemental sulphur seems to play a key role in the formation of pyrite, which is less sensitive to oxidation (unpublished personal experimental results).
Assuming a substantial post-depositional dissolution of autochthonous calcite, pore water in the surface sediments should reflect the Ca/Sr signature of the autochthonous calcite.
Diffusive nutrient exchange across the sediment/water interface is further influenced by a complex of interacting parameters including, e.g. temperature, oxygen availability, vertical and temporal variations in H2S production by SO4 reduction, abundance of bio-degradable organic matter, PO4 retention or release associated with authigenic mineral formation.
After a HNO3/HClO4/HF/HCl-decomposition of 0.25 g solid sample, the determination of major and minor elements, including phosphorus and sulphur, was carried out by sequential ICP-AES (ARL 35000) and external calibration.
Increase of Ca and DIC in the hypolimnion of LGM towards the lake bottom reflects post-depositional dissolution of autochthonous carbonate.
During the exposure of the dialysis cell, changes in interstitial water chemistry can occur, which is why in situ dialysis pore water profiles detected in this manner give an integral picture over the exposure time of the dialysis cells.
Hydrogen sulphide produced by SO4 reduction releases PO4 from its precipitates with Fe. Inflow of SO4-bearing groundwater may therefore indirectly influence the P-cycle of LGM.
Lowering of the pH seems to be sufficient to prevent precipitation of Ca and Mn, which show elevated concentrations in the pore water at the 12 m site.
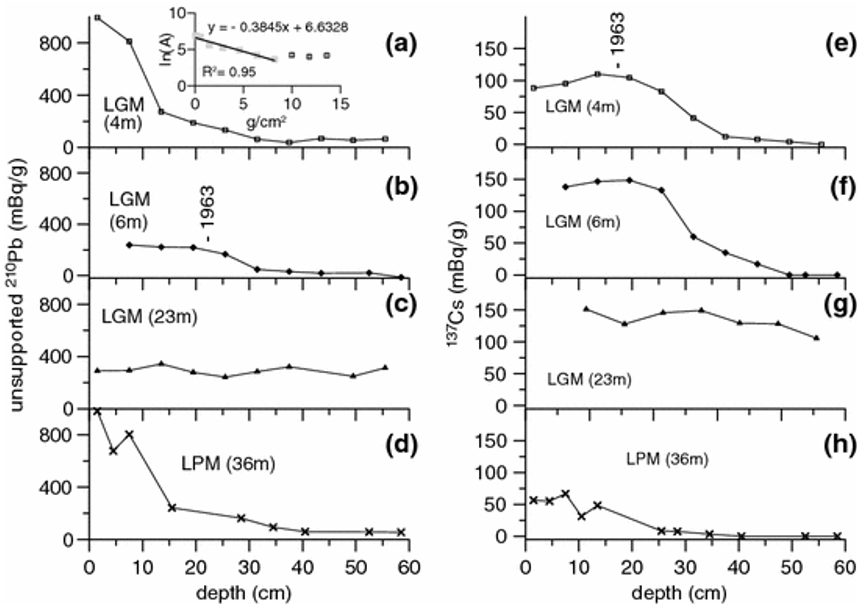
In the lower core section (31.5–55.5 cm) of the 4 m profile, however, unsupported 210Pb values vary between 38 and 69 mBq/g without showing a declining trend versus depth.
If the authors consider TOC and N contents, biogenic opal accounts for approximately 50 wt% in the nearly non-calcareous sediments from the 23 m site.
Three mechanisms may explain the lower Feexc of the profundalLGM sediments: (i) The dissolved Fe influx into the deep water and possible focussing of the settling FeOOH flux towards the centre of the lake basin does not counterbalance the Feexc loss by deposition of FeOOH particles in the shallow water area during overturn.
Because of the permanent absence of oxygen in the LPM deep water, it appears astonishing that OI values of LPM sediments exceed the average OI of LGM sediments by a factor of ~1.5.
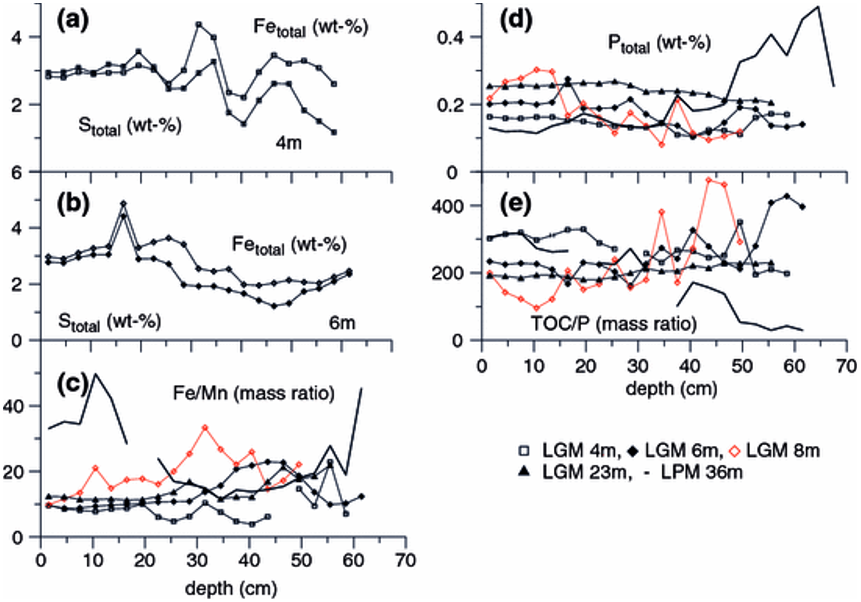
The geochemical signatures of deeper sediments at the 8 m depth, showing distinctly lower Al contents (Fig. 5g), could reflect the presence of minerogenic debris with a geochemical composition that completely differs from those of the other LGM cores.