Marquette University Marquette University
e-Publications@Marquette e-Publications@Marquette
Civil and Environmental Engineering Faculty
Research and Publications
Civil and Environmental Engineering,
Department of
3-12-2019
Adsorption of Organic Micropollutants Onto Biochar: A Review of Adsorption of Organic Micropollutants Onto Biochar: A Review of
Relevant Kinetics, Mechanisms and Equilibrium Relevant Kinetics, Mechanisms and Equilibrium
Yiran Tong
Marquette University
Patrick J. McNamara
Marquette University
, patrick.mcnamara@marquette.edu
Brooke K. Mayer
Marquette University
, Brooke.Mayer@marquette.edu
Follow this and additional works at: https://epublications.marquette.edu/civengin_fac
Part of the Civil Engineering Commons
Recommended Citation Recommended Citation
Tong, Yiran; McNamara, Patrick J.; and Mayer, Brooke K., "Adsorption of Organic Micropollutants Onto
Biochar: A Review of Relevant Kinetics, Mechanisms and Equilibrium" (2019).
Civil and Environmental
Engineering Faculty Research and Publications
. 237.
https://epublications.marquette.edu/civengin_fac/237

Marquette University
e-Publications@Marquette
Civil, Construction and Environmental Engineering Faculty Research and
Publications/College of Engineering
This paper is NOT THE PUBLISHED VERSION; but the author’s final, peer-reviewed manuscript. The
published version may be accessed by following the link in the citation below.
Environmental Science : Water Research and Technology, Vol. 5 (2019): 821-838. DOI. This article is ©
Royal Society of Chemistry and permission has been granted for this version to appear in e-
Publications@Marquette. Royal Society of Chemistry does not grant permission for this article to be
further copied/distributed or hosted elsewhere without the express permission from Royal Society of
Chemistry.
Adsorption of Organic Micropollutants Onto
Biochar: A Review of Relevant Kinetics,
Mechanisms and Equilibrium
Yiran Tong
Department of Civil, Construction and Environmental Engineering, Marquette University, Milwaukee, WI
Patrick J. McNamara
Department of Civil, Construction and Environmental Engineering, Marquette University, Milwaukee, WI
Brooke K. Mayer
Department of Civil, Construction and Environmental Engineering, Marquette University, Milwaukee, WI

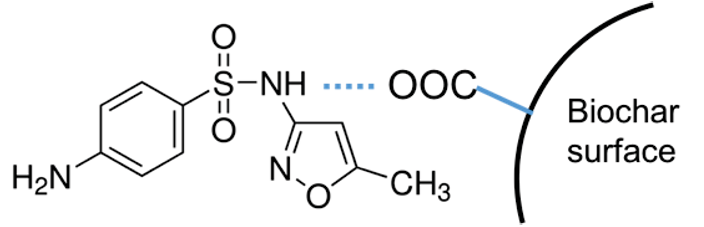
TOC art
Abstract
As an alternative to activated carbon, biochar has been considered for removal of organic micropollutants from
water and wastewater via adsorption. This review elaborates on the fundamental basis of adsorption kinetics,
mechanisms, and equilibrium with respect to biochar-based adsorption of micropollutants. The objectives
include: 1) linking biochar surface properties with adsorption abilities, 2) categorizing the kinetics of adsorption
of aqueous-phase organic compounds onto biochar, 3) categorizing the molecular-scale interactions between
organic micropollutants and biochar, and 4) reviewing existing quantitative methods for characterizing
adsorption equilibrium of organic micropollutants from water onto an adsorbent surface. To fulfill these goals,
the relationships among biochar surface properties, adsorption kinetics, mechanisms, and equilibrium were
clarified as current literature often lacks such discussion or may include conflicting descriptions. Due to its
heterogeneous nature, research on biochar’s adsorption potential for micropollutants is ambiguous. By adapting
adsorption theories to biochar application specifically, this review helps to inform future research in terms of
addressing knowledge gaps in characterizing and improving biochar adsorption.
Keywords
Thermodynamics, sorption, emerging contaminants, wastewater; pyrolysis
Nomenclature
Q mass loading of adsorbate on solids (mg/g)
Q
e
mass loading of adsorbate on solid at adsorption equilibrium (mg/g)
J flux of adsorbate (mg/g/min)
k
f
film diffusion coefficient
A volumetric surface area of adsorbent (m
2
/m
3
)
C adsorbate concentration in aqueous phase (mg/L)
C
e
adsorbate concentration in aqueous phase at equilibrium (mg/L)
D
s
effective surface diffusion coefficient (mm
2
/s)
k
1
, k
2
pseudo first-order (sec
-1
) and second-order rate constants (L/mol/sec)
1.0 Introduction
The occurrence of organic micropollutants in the environment is typically associated with increasing population
and anthropogenic activities
1
. Municipal water resource recovery facilities (WRRFs) are a major source of

organic micropollutant loading to aquatic environments
1
. An estimated 300 million tons per year of
micropollutants, including synthetic industrial chemicals, pharmaceuticals, flame retardants, artificial
sweeteners, and hormones enter natural waters via wastewater discharges
1–3
. Micropollutants are of concern
because they elicit biological and ecological impacts at very low concentrations (ng/L to μg/L)
4–6
.
Micropollutants do not necessarily result in acute toxicity; instead, they are suspected to have long-term effects
on organisms chronically exposed to these compounds
7,8
.
As WRRFs process the majority of wastewater stemming from anthropogenic activities, they are a major
collection and release point of organic micropollutants
3
. The fate of 51 micropollutants during wastewater
treatment is highly variable, as they can be discharged with the liquid effluent, [bio]degraded, volatilized, or
retained on solid surfaces via adsorption, e.g., adsorption to suspended solids, sludge particles, or filter media
9,10
. The adsorptive behavior of organic micropollutants onto solids in engineered and natural environments
substantially affects the fate and removal of micropollutants that are not amenable to biodegradation
11
.
Removal of micropollutants from the aqueous phase during tertiary polishing at WRRFs is commonly achieved
using treatment technologies such as adsorption by porous carbonaceous materials. For example, activated
carbon can remove carbamazepine, 17B-estradiol, and sulfamethoxazole via adsorption
12,13
. Biochar is
another type of carbonaceous solid material, and is produced by pyrolyzing (thermo-conversion under anoxic
conditions) carbon-based feedstock. To volatilize tars, the lower range of pyrolysis temperature is typically 400°C
14–16
. Pyrolysis can produce multiple value-added products such as py-gas, which can potentially offset energy
costs for pyrolysis
17,18
, and biochar, which can be used as a soil amendment and adsorbent
16,19,20
.
Compared to widely-adopted activated carbon, research on biochar as an adsorbent to remove micropollutants
is still in the early stages; however, biochar’s potential for this application is attracting greater attention
21
.
Studies have tested the feasibility of using biochar derived from biosolids, plant residuals, and animal manure to
adsorb hormones, pharmaceuticals, and pesticides
10,20,22,23
. For example, up to 60% adsorption of endocrine
disrupting compounds such as bisphenol A, atrazine, 17α-ethinylestradiol
10
, and triazine herbicides
24
was
observed using plant-derived biochar.
Unlike activated carbon, biochar is produced from more diverse feedstocks under less energy-intensive thermal
conditions. Additionally, chemical activation of the feedstock and gas activation during thermal conversion are
required for producing activated carbon, but they are usually not necessary for producing biochar
25,26
.
Biochar’s unique properties stem from these factors. For example, biochar’s surface area and mechanical
hardness tend to be lower than activated carbon due to production under less energy-intensive thermal
conditions and lack of activation. In spite of the differences, biochar and activated carbon share commonalities
in terms of adsorption functionality, e.g., adsorption via π- π electron-donor-acceptor interactions
primarily relies on the polycyclic aromatic network of the charcoals
27
. Accordingly, the fundamental basis for
adsorption, as described in detail in literature focused on porous adsorbents including activated carbon, is
adaptable to biochar with adjustments in terms of biochar’s properties.
To design and optimize WRRF adsorption treatment processes targeting removal of micropollutants using
biochar, the adsorptive behavior of micropollutants onto biochar must be established. The influence of biochar
surface properties on micropollutant removal is an important consideration for process improvement.

Additionally, adsorption kinetics, mechanisms, and equilibrium are needed to characterize adsorption of
micropollutants from wastewater onto biochar. In the case of aqueous-phase adsorption, kinetics define the
diffusion or reaction rate, reflecting how quickly adsorption occurs. Adsorption mechanisms indicate the type of
intermolecular interactions at play, which depend on the physicochemical properties of the adsorbent and the
adsorbate as well as the bulk solution. Adsorption equilibrium between the solid- and aqueous-phases can be
described using isotherms and thermodynamic parameters.
Adsorption kinetics, mechanisms, and equilibrium are not independent, but relate to one another, making it
important to understand these relationships and their relevance to micropollutant removal efficiency using
biochar. However, previous reviews of aqueous-phase adsorption on biochar have not clearly and consistently
described these relationships
28–32
. Thus, this tutorial review focuses on correlations among the adsorption
concepts of adsorbent surface properties, kinetics, adsorption mechanisms, and equilibrium, in the specific
context of biochar. Equilibrium isotherms provide the boundary conditions needed to solve the diffusion rate
equations characterizing kinetics
33,34
. Equilibrium isotherms are also used to extrapolate thermodynamic
parameters
35–37
. Thermodynamic parameters such as the magnitude of enthalpy change of adsorption
provide good indicators of intermolecular interactions. For example, the enthalpy change of chemical adsorption
involving formation of covalent bonds is on the order of >100 kJ/mol, while smaller changes in enthalpy usually
indicate weak physical adsorption
38,39
. The contribution of adsorption mechanisms to total free energy change
is useful in modifying adsorbent surface properties to improve removal of target contaminants. The rate-limiting
step of adsorption kinetics relates to binding mechanisms as well. For physical adsorption, intramolecular
diffusion is more likely the rate-limiting step, while for adsorption involving shared electron pairs via covalent or
ionic bonds, reaction rate-limiting kinetic models, such as pseudo-first and pseudo-second order (PFO and PSO)
are more appropriate
40
.
It follows that quantitative characterization of a batch adsorption system, such as kinetic and isotherm
modeling, is essential to inform design of flow-through filter beds or column operation
41,42
. However,
misunderstanding fundamental concepts of biochar surface properties and adsorption kinetics, mechanisms,
and equilibrium can lead to inappropriate interpretation and application. Therefore, the goal of this tutorial
review is to introduce and provide a clear overview of the linkages between biochar surface properties and
adsorption potential as well as the theoretical basis and relationships among adsorption kinetics, mechanisms,
and equilibrium. Specifically, this review focuses on clarifying adsorption between aqueous-phase organic
micropollutants and solids materials, which can be applied to biochar adsorption. Critical analysis identifying
knowledge gaps along with research needs to fill these gaps is also presented. Specific objectives include: 1)
linking biochar surface properties with its adsorption abilities, 2) categorizing the kinetics of adsorption of
aqueous-phase organic compounds onto biochar, 3) categorizing the molecular-scale interactions between
organic micropollutants and biochar, and 4) reviewing existing quantitative methods for characterizing
adsorption equilibrium of organic micropollutants from water onto an adsorbent surface.
2.0 Impact of biochar surface properties on adsorption of organic
micropollutants
The adsorption of organic micropollutants onto solid adsorbents is greatly affected by both the nature of the
compounds—e.g., polar vs. non-polar and neutral molecules vs. dissociated ions—and the surface properties of
the adsorbent. For instance, an adsorbent with smaller-sized mesopores can increase steric hindrance for
molecules diffusing into adsorption sites deep in the pores
43
. Additionally, an adsorbent with a high ratio of