




read more










Further studies are needed to explore A. pegreffii distribution in North-East Atlantic and to understand why no Anisakis spp. was found in shads from English and Irish waters.
parasites such as helminths are increasingly used as biological tags to provide information on host populations (e.g. feeding habits, habitat use, stock discrimination, and migration) and on free-living biodiversity and changes in ecosystem structure and functioning (MacKenzie 2002; Marcogliese 2005 for reviews).
Because of their omnipresence and ecological significance, there is a dire need to integrate parasitic helminths in further multidisciplinary investigations to get knowledge on A. alosa and A. fallax and to develop efficient management and conservation programs.
Only one metazoan parasite taxon (i.e. the gill monogenean Gyrodactylus von Nordmann, 1832commonly infecting freshwater fish) has previously been reported in larvae of A. fallax, and four taxa [i.e.
The absence of overlap between parasite communities of immature shads may be due to i) age (larvae, 0+ and older immatures) resulting in potential differences in habitat and food use (Baglinière and Elie 2000), ii) inter-site prey availability inducing differences in the euryphagous and opportunistic diet of immatures (Nunn et al. 2008; Baglinière and Elie 2000), iii) absence of host species needed to complete the heteroxenous life cycle of most helminths, and/or iv) potential mortality of infected immatures as shown for Gyrodactylus (GranoMaldonado et al. 2011).
Chi-square tests and Student t-tests were performed to compare prevalence and abundance of the six common parasite taxa between hosts separately for each of the sympatric sites (Loire, Adour, and North Biscay Bay).
The parasitological parameters used to describe the parasite community structure were: prevalence (P,number of hosts infected with a particular parasite species / number of hosts examined), taxa richness (number of parasite taxa infecting a host species), and abundance (number of individuals of a particular parasite species in/on a single host regardless of whether or not the host is infected) (Bush et al. 1997).
Because helminth parasites are omnipresent in the shads and decrease their fitness, parasitological data must be included in further investigations and management programs on A. alosa and A. fallax.
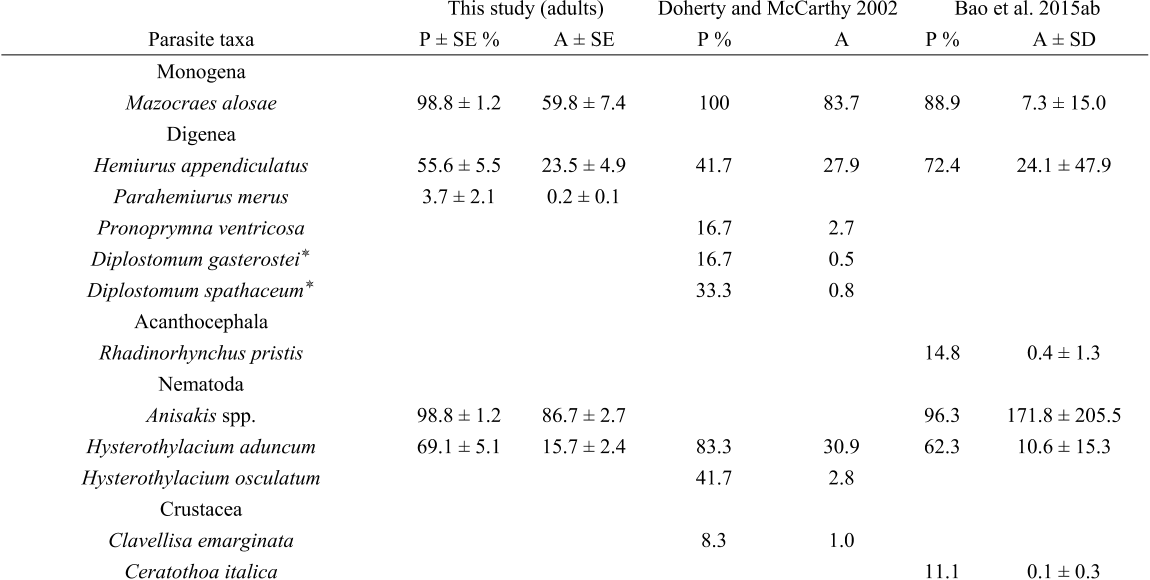
The most prevalent taxa were the monogenean M. alosae (dominant species for A. alosa), the digeneans H. appendiculatus (dominant species for A. fallax) and P. ventricosa (only recorded in A. fallax), the cestode E. fragile (rare in A. alosa), and the nematodes Anisakis spp. (i.e. A. simplex s.s., A. pegreffii) and H. aduncum.
Because greater pathogenicity is often observed for recent host-parasite associations (e.g. Kennedy 1994; Kania et al. 2010), the association between H. appendiculatus and A. alosa could be more recent, thus inducing a significant host fitness loss.
In models analyzing all individuals, explanatory variables considered were the host species, the sympatry (yes/no) and the interaction between these two factors.
Their study highlights the importance of metazoan parasites in A. alosa and A. fallax in terms of theirtotal prevalence of 100% and their high mean abundance (respectively 167 ± 10 and 112 ± 11 parasites per fish) and diversity (nine parasite taxa per host species).
All the metazoan parasites found were numbered per organ and per fish, and morphologically identified to the species level excepted for nematodes.