




read more









skeletal muscle differentiation in the human does continue after birth with up to 20% of fibres undifferentiated in the newborn, which drops to adult levels by 1 year of age.
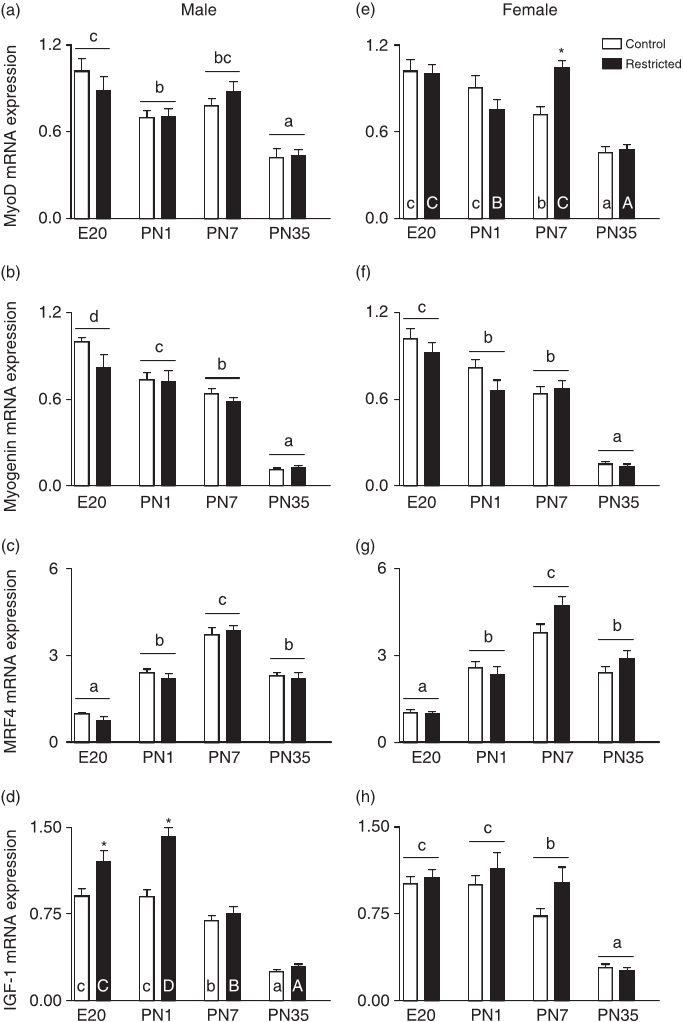
Their laboratory has shown that foetal growth restriction reduces gene and protein markers of skeletal muscle mitochondrial biogenesis [e.g. peroxisome proliferator-activated receptor (PPAR)-g coactivator-1a (PGC-1a), mitochondrial transcription factor A (Tfam), cytochrome c oxidase subunits 3 and 4 (COX III and IV)] in 6-month-old adult rats, with males more affected than females.
Their laboratory has shown that uteroplacental insufficiency in pregnant rats not only impairs the growth of the foetus but also impairs maternal mammary development resulting in poor lactation,40 further compromising the growth of the offspring after birth.
One study reported that the master regulator of mitochondrial biogenesis, PGC1a, mRNA was downregulated in the slow twitch soleus muscle but upregulated in the fast twitch extensor digitorum longus muscle of small birth weight offspring at 21 days of age, with males more affected than females.
Cross-fostering Restricted pups onto a Control mother significantly increased COX III mRNA in males and COX IV mRNA in both sexes above controls with little effect on other genes.
Developmental age appears to be a major factor regulating skeletal muscle mitochondrial and developmental genes, with growth restriction and cross-fostering having only subtle effects.
Analysis for studies 1 and 2: real-time polymerase chain reaction (PCR) analysisFor the Developmental Timeline Study, total RNA was extracted from skeletal muscle using the Tri-Reagent (Ambion Inc., Austin, TX, USA) method due to the small amounts of pooled tissue obtained from pups at E20 and PN1.
How this will impact on the adult skeletal muscle metabolic profile is unknown, but as cross-fostering has been associated with improved glucose tolerance in adulthood of small birth weight rats,45 the gene changes in skeletal muscle may also contribute to improved adult health.
Whether these relatively minor gene changes following cross-fostering will contribute to later disease prevention, observed at 6 months of age,45,46,51 is unclear; but perhaps an intervention that occurs at an age when deficits in skeletal muscle mitochondrial biogenesis markers are present would provide greater benefits to improved skeletal muscle mitochondrial biogenesis and insulin sensitivity.
more recently Costello et al.24 reported that following late gestation undernutrition in sheep, reduced myofibre density was associated with a compensatory upregulation of the IGF-I receptor at127 days gestation (term 147 days).
In low birth weight humans, improved growth between birth and 2 years has been shown to reduce the risk of developing adult metabolic disease.
On the basis ofour and others findings in adulthood, the authors hypothesized that key genes involved in skeletal muscle development and mitochondrial biogenesis would be altered following intrauterine growth restriction in rats, with different developmental profiles between males and females.
It is important to consider that the control group used in these studies had their litter size reduced at birth,10,11 previously shown to alter postnatal growth and impair skeletal muscle markers of mitochondrial biogenesis at 6 months of age8 and therefore impacts on the interpretation of these findings.
It was particularly surprising to find that foetal growth restriction had no impact on PGC-1a gene expression as uteroplacental insufficiency causes foetal hypoxia and in skeletal muscle the hypoxia inducible factor 1 activity and expression is tightly coupled to PGC-1a.60